ANAGENESIS AND MACROEVOLUTION
This is my Ph.D. dissertation, a footnote to Darwin’s Origin of Species, dealing with Origins of higher ranking groups, especially Phyla and Superphyla.
ANAGENESIS AND MACROEVOLUTION
THE ORIGIN OP HIGHER TAXA PROM LOWLY ORGANIZED ANCESTORS, WITH A DISCUSSION OF THE PHYSICAL BASIS
OP THE RISE OF BIOLOGICAL ORGANIZATION
A DISSERTATION SUBMITTED TO THE DEPARTMENT OF BIOLOGICAL SCIENCES AND THE COMMITTEE ON THE GRADUATE DIVISION OF STANFORD UNIVERSITY IN PARTIAL FULPILLMENT OF THE REQUIREMENTS
FOR THE DEGREE OF DOCTOR OF PHILOSOPHY
By John Francis Bennett
December 1961
Approved for the University Committee on the Graduate Division:
Dean of the Graduate Division
ACKNOWLEDGMENTS
In supporting the development of this theoretical work as a doctoral dissertation, the Department of Biological Sciences of Stanford University has undertaken an unusual responsibility. I acknowledge my debt for this support, and recognize my own responsibility in consequence. The burden has seemed heavy at times, notably because there is no field among the several which bear on this work, in which many others are not in closer touch with the facts than I. I hope for the reader’s forbearance, and indeed more, I hope that where he can teach me better he will do so.
My deficiencies of knowledge and understanding have been lessened by members of the department’s faculty, students, and members of other departments of the University, through discussions of ideas and critical reading of preliminary manuscripts over a period of years. I wish to thank them all, though I cannot name them all.
I am especially indebted to my advisors on the completed manuscript, Professors Abbott, Giese, Holm, Regnery, and Danforth. Each has contributed general appraisal and criticism from the vantage point of a different discipline, in addition to making valuable suggestions on points of fact and details of expression. Important expansions and clarifications of several of the more abstract discussions in the work have resulted from Professor Abbott’s final critical reading of the manuscript as principal advisor; I am deeply obliged to him for the great pains he has taken. Special thanks go also to Professor F. O. Koenig of the Department of Chemistry at Stanford, for the critical reading and enjoyable discussion of Part II. Finally,. I should like to express special acknowledgment of my debt
to the late Professor G. F. Ferris, who originally sponsored this work and encouraged its development.
TABLE OF CONTENTS Page
INTRODUCTION 4
PART I. THE ROLE OF ANAGENESIS IN
THE MACROEVOLUTION PROBLEM
Section 1. TAXONOMIC TERMINOLOGY. 6 2. BACKGROUND 7
3. GOLDSCHMIDT’S TAXONOMIC ARGUMENT 11
4. CRITERIA OF TAXONOMIC RANK 14
a. Divergence
b. Diversity
5. THE FORMATION OF HIGHER TAXA. 16
6. THE CRITICAL QUESTION. 17
7. – DISTINGUISHING CHARACTERS 19
a. Role of gaps
b. Role of extinction
c. The non-extinction of phyla
8. DISTINGUISHING CHARACTERS OF HIGH
RANKING TAXA
9. THE ROLE OF ANAGENESIS
10. A DIAGRAMMATIC REPRESENTATION
OF THE ANAGENETIC VIEWPOINT
11. THE GENETICS OF ANAGENESIS
a. An idealization: the “gene tree”
b. A note on recapitulation
c. A return to the problem of new genes
12. LONG TERM IMPLICATIONS OF ANAGENESIS
a. “Dynamic taxonomy”
PART I I . INQUIRY INTO THE PHYSICAL MEANING OF ANAGENESIS
Section
13. DEFINITIONS
14. “CLASSICAL” MANIFESTATIONS OF ANAGENESIS
15. THE BEARING OF ANAGENESIS ON
BIOLOGICAL THEORY
16. A PHYSICAL INTERPRETATION OF ANAGENESIS .
17. APPLICATIONS TO BIOLOGY.
a. The evolution of. exploitation of phenomena
b. The evolution of organ systems
c. The evolution’ of ecosystems
LITERATURE
LIST OF FIGURES
Figure
1. A phylogenetic “gene tree”
2. Average relative ages of taxonomic categories
3. Diversification through natural selection, as illustrated
in the Origin of species
INTRODUCTION
This theoretical dissertation stems from reflection on an argument of the late Richard B. Goldschmidt. Briefly
adumbrated, the argument is that high ranking taxa must have arisen by means of saltation, because their great differences from one another were present from their beginning. The argument seems to me erroneous, but not wholly so. It served to suggest to me that the great differences between the phyla should be due in part to their origin from relatively simple ancestors.
In the words of Professor C. H.Danforth (unpublished), who conceived this answer to Goldschmidt independently and many years before me, siblings might have been “in different phyla,” i.e., might have been the progenitors of different phyla, in a population of very primitive organisms in remote past times. In such populations slight mutations in different directions could be expected to cause variations in structure or function which laid the foundations for what we recognize today as characters of different phyla. With advance in level of organization, in later eras, this expectation should decrease, since slight mutations would tend to lay the foundations for
relatively superficial characters in more complex organisms.
Part I is concerned primarily with the development of
this approach to ihe macroevolution problem: the problem of the mechanism of origin of high ranking taxa. While considerable space is devoted to the restricted objective of meeting Goldschmidt’s argument, the development of the approach has the more general objective of contributing to the advance of the modern synthesis in evolution theory. The synthetic theory rests on the hypothesis of “quasi-continuous” evolution, by whic.h I mean the hypothesis that
offspring have ‘virtually always closely resembled their parents, even though their variations have presumably involved discontinuous events at the molecular level. Goldschmidt’s saltation hypothesis is a contrasting alternative, which is argued here to be unnecessary. The factor of evolutionary rise of level of organization of living matter, although universally recognized, has implications which have not been stressed in recent discussions of macroevolution. Its bearing on Goldschmidt’s argument may be regarded as one such implication. Others are discussed in the last two sections of Part I, in relation to the problem of new genes, and to the problems for theoretical taxonomy posed by the indefinite growth of the tree of life.
The diagram presented in section 10 provides a visual summary of some of the discussion centered on Goldschmidt’s argument, and at the same time provides a frame of reference for the subsequent discussion of genetics, “dynamic taxonomy,” and related topics.
For the general advance in level of organization of organisms and of ecosystems characteristic of evolution as a whole, I have adopted the term anagenesis, after Rensch (1947 et seq.) and Huxley (1957).
My efforts to understand the physical basis of anagenesis, thus broadly conceived, have led to Part II of this work. Some embryologists and philosophers, as well as evolutionary biologists, have discussed the-basis for the remarkable phenomena wherein
increasingly highly organized and improbable states of matter and energy make their appearance in nature. On the whole, however, the solid contributions have been of a descriptive nature, and the phenomena have yielded little to theoretical illumination, with the result that discussion of them from the standpoint of general principles has tended to fade into the scientific background.
I have sought the bearing of physics and molecular biology on the understanding of anagenesis, drawing especially upon the writings of the physicists Schrödinger, Weizsacker, Brillouin, and Rothstein, while trying to take much else also into account.
What is submitted here is largely exploratory and tentative. It is presented with the hope that the approach may lead to a deeper level of comprehension of biology and man, as a step towards the synthesis of life science with physical science to the extent that such synthesis is possible in principle. Something will be gained if the approach is found interesting, and as much as is aspired to will be gained if authorities in various chapters of phylogeny, and even in certain widely different areas such as economic history and neurophysiology, find it worth while to look at their specialties in a
similar way, more closely than I am able to do.
.
PART I. THE ROLE OF ANAGENESIS IN THE MACROEVOLUTION PROBLEM
1. TAXONOMIC TERMINOLOGY.
The recent book on Principles of animal taxonomy by G. G. Simpson (1961) provides an authoritative and convenient point of reference for following sections . A few of Simpson’s (1961:19) most fundamental definitions are repeated here, for purposes of future discussion. Undefined terms on which these definitions are based, and other taxonomic terms used in this work, are discussed by Simpson. They are not examined here on the grounds that they are largely self-explanatory, and that Simpson’s work is available for reference.
A taxon is a group of real organisms recognized as a formal unit at any 1eve1 of a hierarchic classification. A taxon is thus a population of objects. Fossils are included; the problem raised by their fragmentation does not concern us. A taxonomic category or (in the present context) simply a category is a class the members of which are all the taxa placed at a given level in a hierarchic classification.
The rank of a category is either its absolute position in a given hierarchic sequence of categories or its position relative to other categories. These alternatives can be distingished by simply adding the adjective “absolute” or “relative,” when necessary.
The rank of a taxon is that of the category of which
it is a member. Simpson employs Beckner’s (1959) symbol
for a taxon of rank Tj (The corresponding symbol for a
J category of rank j is Cj .) Ranks run from j = 1 (lowest) to
j =n (highest); n may vary from as low as 7 up to about 30, at the discretion of the individual taxonomist.
It is advisable in our theoretical discussion to depart somewhat from Simpson’s concept of “taxon.” Although Simpson insists upon monophy1y as a criterion, he defines monophy1y so broadly (descent from an immediately ancestral taxon of equal or lower rank; 1961:124) that, in effect, a considerable measure of polyphyly may inhere in a population of organisms which satisfies his definition of taxon.
Objections to so broad a concept of monophyly and to its taxonomic results have been raised (cf. Reed 1960, van Valen 1960, Emerson 1961), but practical considerations provide Simpson with a good case. It is not to the point to enter into this dispute. The advantage for present purposes, however, goes with taking a more idealistic view of “taxon,” in line with that of Reed, viz., as the population consisting of a common ancestor and all its descendants, where “common ancestor” means the most recent gene-exchanging unit which includes all or nearly all the genetic stock of the population. It is understood that this unit may run in size from a single organism to a pair, deme, species, or even (in some plants, in earthworms, weevils, brine shrimps, etc.) polyploid complex, but that, so far as we know, it does not involve categories higher than family and rarely involves different genera. In practice it is virtually never known what the unit is, especially at the higher taxonomic levels which are our main concern here. The over-all success of the group-within-group scheme of classification is a convincing indication that anastomosis of gene pools has not frequently occurred across generic or familial boundaries, however.
In principle it might be preferable to use “clade” rather than “taxon” for my concept (cf. Huxley 1957), reserving “taxon” for the classifiers’ actual groups, in accord with Simpson’s definition. As Huxley points out, named taxa may be either “grades” (groups of forms of a given kind and level of organization) or “clades” (mono- phyletic groups in the narrower sense) or both, while our concern is with the latter. What the evolutionary classifier aims at, however, is a grouping primarily by clades, insofar .as possible within the limits posed by the fact that sequences blend as the gaps in our knowledge are closed. Hence, no important distinction is confounded, at least the context of this work, by using the more usual term for the idealistic concept. Also, certain terminological inconveniences are avoided by so doing.
2 . BACKGROUND.
The taxonomic argument which initiated this work is considered at some length in the next section. It may be questioned whether the argument merits so much consideration, since it rests in part on a mistake which could have been avoided on the basis of earlier discussions of taxonomic principles (cf. Shull 1936, 1951), and which perhaps now, on the strength of Simpson’s discussion
(1961:69, 142), should be considered impossible to a well-informed evolutionist. My reasons for going into the matter are the following. First, I believe Goldschmidt was not entirely wrong, else there would be nothing to contribute here. Second, the error is not so obvious that it cannot bear reconsideration. Third, it is often worth while to dwell on the thought of a man of Goldschmidt’s eminence even where it
is wrong. Goldschmidt was described by Wright (1959), himself a
geneticist noted for laying foundations for the modern synthesis in evolution theory, as “a very great geneticist.” As an evolutionist Goldschmidt has not this stature, but it is noteworthy that he was among the first to foresee the possible synthesis of Mendelian genetics and Darwinian selection theory, and it was this perception which seems to have motivated his study of the genetics of races .of the gypsy moth, Lymantria dispar, which remains one of the most thorough and comprehensive studies in experimental population genetics. His most significant work, on the genetics of sex and development, may in fact be regarded as a detour from his primary concern with the relation of genetics to evolution. It is of interest to understand how a man ahead of his time in pursuing the synthesis of Mendelian and Darwinian ideas, was led to dispute vehemently the adequacy of the synthesis which ultimately was claimed.
Goldschmidt’s last (posthumous) published words on the question were (1960:307):
The material basis of evolution . . . put a finger on some major errors in present day evolution theory. Though it is still violently attacked today by the partisans of one very fashionable way of looking at evolution, I am certain that in the end I shall turn out to have been right.
It is not impossible that there is a relation between the taxonomic argument here at issue, and Goldschmidt’s trenchant convictions, this argument being the only one not adequately countered by his opponents, to the best of my knowledge. The attacks of which Goldschmidt speaks are no longer violent, if that is indeed the word. There are not a few evolutionists who feel that the whole issue has been, or should have been, long since laid to rest. But there have been positive defenders of the saltation viewpoint other than Goldschmidt in recent years, notably Schindewolf (see 1950), as well as some who have leaned in Goldschmidt’s direction to varying degrees, on the basis of very different classes of facts (e.g., Petrunkevitch 1952; Beerbower 1953; Flower 1955; Lewis and Raven 1958; Brough 1958; Dodson 1960).
Thus the controversy which he fomented is still a matter of
interest. Goldschmidt kept this controversy alive, against strengthening opposition from proponents of the neo-Darwinian or synthetic theory, throughout his last 25 years (1933-58). His quarrel was with the theory’s adequacy to explain, in terms of cumulative action of selection on slight variations, the origin of taxa at and above the rank of species, i.e., to explain “macroevolution” as he conceived the term. He did not question its adequacy so to account for “microevolution,” or the deployment of a species into new areas and climates, with the formation of corresponding appropriately adapted races and subspecies
The terms “micro-” and “macroevolution” seem to have been introduced by Dobzhansky (1937). Rensch (1947 et seq.), because of the linguistically hybrid nature of these words, has instead used “infraspecific” and “transspecific evolution” in his distinguished book, but usage is such that reform seems unlikely. The concepts have survived a measure of criticism. Simpson (1944) introduced “megaevolution” to denote the evolution of new classes and still higher taxa, thereby restricting the coverage of “macroevolution” roughly to categories between species and class. There has not proved to be much advantage in this refinement, but it is worth noting here that it is largely the phenomena of “megaevolution,” by this definition, which come under consideration in this work.
Later (1953) Simpson emphasized the artificiality of all these distinctions, and suggested that each taxonomic level may present its own problems of evolutionary explanation, so that one might treat separately the origin of orders, families, etc., with as much justice
as micro-, macro-, and mega-evolution. But subsequently (1959a; see also 1961) Simpson showed that quite different types of evolutionary pattern are found at each level at least between family and class, so that it is rather as if every taxon presents a unique problem, except that certain patterns frequently recur. The fact that ranks of taxa are somewhat arbitrary and subject to revision in the classification of any particular group of organisms does not, of course, aid in the identification of such characteristic patterns as may exist at different levels.
They were, rather, scientifically interpretable, through his own theory of physiological genetics, in terms of altered rates of gene-controlled chemical reactions, especially in early development. He pointed to homoeotic mutants as examples of the reality of his concept of systemic mutation, and studied them especially in Drosophila.
Thus Goldschmidt developed a “saltation” hypothesis or
theory of macroevolution. The general idea is an old one, going back at least as far as Kalliker and T. H. Huxley (cf. Singer 1950:306), but at Goldschmidt’s hands it probably reached its highest ascendance, because of the revitalization he was able to give it with an erudite modernization, energetically pursued. The idea has, all the same, never carried conviction widely. The principle reason for this is probably the intuition that a “hopeful monster” is analogous to a scrambled watch, and to the extent that the living organism is a more intricate mechanism than a watch, it is even less likely that a hopeful monster will succeed than that a scrambled watch will be a useful mechanism of any sort. This objection can be answered with the familiar argument that in sufficient time, a monkey at a typewriter will inevitably compose the complete works of Shakespeare. But an answer which does not depend on so great a concurrence of chance events is to be preferred as less improbable, especially if its plausibility is otherwise equal or greater.
3. GOLDSCHMIDT’S TAXONOMIC ARGUMENT.
The first half of The Material Basis of Evolution is devoted to the presentation of a number of arguments against the validity of the neo-Darwinians’ implicit assumption of quasi-continuous evolution in the origin of “good” species and transspecific taxa. Each argument has been met in one or more of the standard treatises which together constitute the basis of the modern synthetic theory of evolution, notably Dobzhansky (1941; the first edition of 1937 predates Goldschmidt’s book), Huxley (1942), Mayr (1942), Stebbins(1950), Simpson (1944,1949,1953), and Rensch (1947 et seq.); see also Simpson (1960a) on the especially critical question of gaps in the fossil record.
The “taxonomic” argument which Goldschmidt presented in 1952 appears to have been an afterthought, but may have been a motivating factor from the outset. Its essence is contained in the following passages (1952:91-2):
Looking at the taxonomic order of animals and plants, we see the picture represented as a pedigree or tree of descent. This means that a phylum consists of a number
of classes all of which are basically recognizable as belonging to the phylum but, in addition, are different from each other. The same principle is repeated at each taxonomic level. All the genera of a family have
in common the traits which characterize the family; e.g., all genera of penguins are penguins. But among themselves they differ from genus to genus. So it goes on down to the level of species. Can this mean anything but that the type of the phylum was evolved first and later separated into the types of the classes, then the orders, and so on down the line? This natural, naive interpretation of the existing hierarchy of forms actually agrees with the historical facts furnished by paleontology. The phyla existing today can be followed farthest back into remote geological time. Classes are a little younger, still younger are the orders, and so on until we come to the recent species which appear only in the latest geological epochs. Thus logic as well as historical fact tells us that the big categories existed first, and that in time they split in the form of the geneological tree into lower and still lower categories. But the Neo-Darwinians teach just the opposite. By accumulation of small genetic differences subspecies are transformed into species; more accumulation of small differences in a species makes it acquire generic rank; the genera repeat the process to develop slowly into families, and so forth until a phylum has been separated.
Thus we are presented with the following picture. As an example, let us consider the origin of birds. In the evolution of reptiles, by slow accumulation of mutants, some reptile subspecies became a species; from this species a genus was derived in the same way, with characters leading in a definite direction. Within this genus the subspecies of one species could be called the representative of a new family. This new type now had to form new subspecies which transformed slowly into different species, and these again into genera, from which by the same process maybe another family was formed, which led still farther away in the direction of what was to become a bird. The same process finally led to orders, from one of which again by the same process a representative of a new class, the birds, derived. After this method, which I should like to call the “crazy quilt” method, had produced the first bird, the whole story would have to begin again in the opposite direction. Although the first bird had evolved by the divergence of more and more accumulated mutants along some reptile line, this new form must then have started to build up subspecies which diverged into species and so on until all the orders, families, genera, etc., of birds had been diversified. But all of them remained birds, while formerly the same process had made birds out of reptiles. Such are the workings of evolution according to the Neo-Darwinian scheme!
It is hardly surprising that a number of zoologists, botanists, and paleontologists were not convinced that such a scheme could work. Certainly the evolutionary steps leading from a reptile to a bird should be infinitely more numerous than those leading from one kind of bird to another. But neither in this nor in any comparable case of macroevolution has more than an indication of these series been found.
The answer to this argument, in brief and partial summary, is as follows: While it is true that, at least as a ,rule, a phylum is older than its classes (it is of course never younger), it does not follow that the phylum existed first, because any taxon begins as a single gene-exchanging unit not often higher than species in rank, and achieves genus, order, phylum rank, etc., only as it branches and rebranches in the course of its adaptive radiation. Even if the ancestral unit were a “hopeful monster” characterized by a systemic mutation, this would be true, subject to the proviso that an essentially monotypic line may deserve high taxonomic rank after a time much longer’ than that of the average life-span of species, if its divergence in organization from nearest related taxa is as great as that of polytypic groups generally ranked correspondingly high.
If the following principle be invoked, the saltation hypothesis is not necessary to explain such tendency as may exist for older monotypic lines to show greater divergence from adjacent taxa than do more recent lines: As a rule, the lower the level of organization of the ancestor, the higher the relative rank which may be achieved by the highest and most inclusive taxon to which it gives rise, i.e., the greater its “taxonomic potentia1.” This rule is important for the understanding of the divergence of polytypic as well as monotypic taxa, hence for the understanding of the origin of high ranking taxa in general.
4. CRITERIA OF TAXONOMIC RANK.
The discussion here should be considered supplementary to that of Simpson (1961, chapters 4 and 6), although I have tried to make it largely self-contained.
A population of organisms is recognized as a taxon on the basis of similarities which are consistent with the criteria of a) monophyly, and b) gaps with respect to other populations. Once recognized, the principle criteria for fixing the rank of the taxon are divergence and diversity. Other factors, which will not be discussed here, include relative antiquity, and convenience. In order to be able to extend the potential application of the former concepts to very high categories, I shall use somewhat different definitions of divergence and diversity than those implicit in Simpson’s discussion (1961:132-4).
a. Divergence. Instead of only the sum of dissimilarities in homol-
ogous and to some extent in parallel characters but no others, I take divergence to include also and primarily the relative proportion of non-homologous characters. In comparing a tunicate with a vertebrate, for instance, an “index of divergence” could in principle be formed as the ratio of characters such as backbone, eye, tunic, and blood-flow reversal, found in one form only, to the totality of characters, including notochord, gill slits, and nerve tube found in both. The common characters should be included twice in the totality of characters, once for each appearance. The complementary proportion (one minus the index of divergence) would of course be the ratio of (twice) the characters-in-common to total characters, and would constitute an “index of homology.” This term is appropriate, since similarities attributable to analogy and convergence should be tabulated as differences. The utilization of degrees of similarity of the homologous characters, after the manner of Michener and Sokal (1957) for instance, would in effect reduce the factor of two by which the characters-in-common are entered. Actual application of such indices may be in the rather distant future, principally because there is great disparity in the relative complexity of, and information about,
“characters.” The concepts are, all the same, quantitative in principle, and have the advantage over other quantitative concepts of similarity and dissimilarity in allowing for the inclusion of all information about the organisms, or more broadly, life cycles, under comparison.
From the standpoint of the genetic bases of characters, the index of homology may be regarded as an approximate “index of genes-in-common,” or at least of genes-once-in-common, while the complementary index of divergence provides an estimate of the proportion of genetic material of the combined descendant forms not represented in the common ancestor.
This genetic interpretation is presented only as a guide to thinking, there being as yet little evidence for deciding its validity and utility. The indices have been defined with reference to a pair of given organisms, or better, of given life-cycles, it being life-cycles, and forms representing stages of them, which nature presents for investigation. Indices of divergence and of homology between entire taxa could be defined as average indices taken over all possible pairs of individuals which include one member from each taxon. This is
of course a totally impractical definition, but one in terms of an unbiased sample of such pairs can be made, based on the same idea. It is in this sense that future reference to the “degree of divergence” between taxa should be understood.
It is not to be thought that the index of divergence for any two given forms would be a perfectly accurate indication of either remoteness in time of their descent from a common ancestor, or of the amount of genetic material that they hold in common. Bigelow’s (1956) example of bird, crocodile, and lizard attests to the contrary, unless appearance be remarkably deceiving in respect to characters-in-common between crocodile and lizard. That the index would be strongly correlated with these factors, however (positively in the first case and negatively in the second), is indicated by the consensus that dissimilarity in general is so correlated (cf. Bader 1958; Simpson 1961:192).
b. Diversity. . In order to free the concept of diversity from any .
connection with dissimilarity, hence with divergence, it is necessary to imagine, for the moment, that the criteria of monophyly and gaps which mark off taxa can be judged from pedigrees alone; also that “polytypy” implies only a multiplicity of genetically isolated groups, and carries no connotation of dissimilarity. Simpson conceives diversity of a taxon as the extent to which it is polytypic (1961:134). For present purposes it is desirable to distinguish between breadth and depth of diversity, or synonymously between lateral and vertical diversity, respectively.
I take extent of polytypy as a measure of breadth of diversity, and number of nestings of groups-within-groups, i.e., number of levels of subsidiary taxa, as the measure of depth. With respect to lateral diversity, Simpson conceives the extent of polytypy to be determined by the number of Tj-1s included in a Tj. Perhaps a better indication of lateral diversity of a taxon is given by the number of species which it includes, i.e., the number of Tjs rather than T j-1s (disallowing the inclusion of subspecific taxa in the Tj notation). Still better might be the number of all included taxa, from Tj-1s to Tjs. It is not a matter of practical importance to decide this question here. In what follows, one of these criteria is perhaps as good as another for what is meant by lateral diversity, with number of species to be taken as ruling in the event of ambiguity, on the basis that this is at least as good a criterion as Simpson’s, and is more familiar.
It is depth of diversity, however, which is more significant as a criterion of taxonomic rank. Each recognizable sub-group level requires, at least ideally, a separate rank, so that if there are, say, 6 levels of sub-groups, the whole group should be a T 7, whereas another taxon with only 3 levels of sub-groups should be only a T 4 even though it may contain many more species, therefore more
breadth of diversity. This argument is subject to the proviso, however, that divergence overrides vertical as well as horizontal diversity in the determination of rank.
5. THE FORMATION OF HIGHER TAXA.
It is obvious that without diversification there would be no higher taxa. Neglecting divergence, vertical diversification operates as follows in their formation.
A given species splits up into a group of species by undergoing familiar microevolutionary processes; this results in the formation of a genus, assuming that “genus” means “group of closest related species.” Another splitting by some or all of the species of the group begets a group of groups of species, which is a family if that rank be so defined; etc. Note that the genus is older than its species (other than the species of the common ancestor), and the family older than its genera, etc., age being measured from the time of existence of the common ancestor; but the genus, family, etc., all began with a single gene-exchanging unit (taken as a species here, for simplicity) and did not come into existence as taxa of higher rank except as an increased number of nestings of groups within groups was formed, i.e., until a requisite measure of vertical diversification had occurred. It follows that from the standpoint of diversification, a taxon gains rank as time passes; hence, from this standpoint it is incorrect to say, as Goldschmidt does, “Thus logic as well as historical fact tells us that the big categories existed first . . . ”
It is equally obvious, however, that without divergence there could be no higher taxa, regardless of diversification, unless pedigrees were known with considerable precision, because all living things would then look alike. Moreover, divergence tends to dominate diversity as a criterion of taxonomic rank. This is indicated by the fact that such diversified groups as the higher insect orders are ranked well below some groups with very few species and correspondingly few nested sub-groupings, e.g., Mesozoa, Tardigrada, Psilopsida; by the recognition of mono- or oligotypic groups at every taxonomic level; and by the history of taxonomy, which, for example, shows a progressive demotion of Linnaeus’
“classes” of flowering plants relative to the cryptogamic groups, and of the vertebrate classes relative to the “Vermes,” this demotion being primarily the result of the much greater divergence found among cryptogams and invertebrates when these groups were subsequently studied more closely. This historical trend could conceivably continue, incidentally, with the elevation of various moneran groups to the rank of kingdom as their divergence, primarily biochemical, becomes better known. On the other hand, anastomosis of gene pools might prove to be so extensive among monerans that a phylogenetic classification is impossible in principle.
6. THE CRITICAL QUESTION.
In general, the higher the rank of a taxon, the greater its divergence from systematically closest related taxa. This generalization permits a statement of the critical question implied in Goldschmidt’s argument: Was the greater divergence between higher taxa already established in their respective common ancestors, i.e., were these ancestors already as divergent from nearest relatives as are their descendants, as groups, or has the divergence merely been accumulated between their descendants, as groups, with the passage of time? The contrasting macroevolutionary viewpoints at issue in this work present correspondingly contrasting answers to this question:
- The saltation viewpoint. Although he does not arrive at the critical question as such, Goldschmidt (1952) in-effect answers that the greater divergence was there to start. His explanation, in keeping with his previously developed theory, is that the common ancestors of the higher taxa were hopeful monsters resulting from systemic mutations. It would seem to follow that the greatest saltations occurred earliest to produce the kingdoms, then at a later time saltations of a second order of magnitude occurred to produce the phyla, etc., although Goldschmidt avoids this conclusion.
ii. The synthetic viewpoint. The greater divergence was built up gradually. The following quotation from Simpson (1953:342-347) represents to the best of my knowledge the most nearly direct answer to the critical question which has been advanced by proponents of the synthetic theory:
A higher category [taxon] is higher because it became distinctive, varied, or both to a higher degree and not directly because of characteristics it had when it was arising. [A comparison of recent vs. early Paleocene ungulates and carnivores is made to illustrate this point.]
. . . The few characters that are common to the ancestral and most descendant forms, such as large canines and ” brachydonty in carnivores, are merely primitive characters for most or all placental mammals. In the early paleocene carnivores these are not new characters the rise of which produced an order Carnivora, and there are no such characters. Moreover, the characters-in-common of ancestral carnivores (creodonts) and ungulates (condylarths) in the early Paleocene are exactly the same in both groups. It is most unusual to get good samples of the ancestors of higher categories so near their point of divergence. The example proves beyond possible doubt that in this case, at least, the higher categories did not arise as’such, that there was no “archetype,” no “generalized” ancestor with the characters-in-common of the order, no “systemic mutation,”. . .The characters that distinguish higher categories are adaptive and they are the same sorts of characters, although often cumulatively greater in degree, as adaptive characters involved in speciation.
iii. The anagenetic viewpoint. A higher taxon which includes complex forms is higher in part because its common ancestor was a simpler form. Its greater divergence was built up gradually, but there was a greater potential for divergence to start.
The anagenetic viewpoint is not as radical a departure from the synthetic as the saltation viewpoint is. It may indeed appear to involve a modification of the synthetic viewpoint too minor to be of any appreciable significance, the more so as there is nothing in it which is not already implicit in the working hypotheses of evolutionary biologists. Yet, if it is worth while to attempt to answer Goldschmidt and fully reconcile the macroevolution controversy, this modification appears to be essential.
To show that anagenesis has an essential bearing on the macroevolution controversy is to make use of it actively in evolution theory, rather than passively as heretofore. Since anagenesis is anything but a minor factor in the whole sweep of evolution, a view of macroevolution which does not make active use of it is incomplete. Here, perhaps, is the most general and significant advantage which the anagenetic viewpoint has over the synthetic, from the purely theoretical standpoint.
7. DISTINGUISHING CHARACTERS.
While Goldschmidt’s inference that “the big categ6ries existed first” is not entirely correct for reasons given, and leads to a faulty description of neo-Darwinian teachings of the evolution of higher taxa insofar as that description misrepresents the role of diversification, his implication that the type of the big categories existed first is not without a degree of validity.
For “type” we may substitute “distinguishing characters” without fundamental distortion of Goldschmidt’s thought. The term “distinguishing characters” refers to characters peculiar to the members of a particular group, as, for example, the primary possession of nematocysts is peculiar to the members of the phylum Coelenterata. (Evidence of the production of nematocysts by one species of ctenophore has, however, been reported; see Hyman 1959:730.) “Characters-in-common” is perhaps usually used in the same sense, but this usage is open to the objection that members of a given order, say, have in common not only the distinguishing characters of the order, but also the characters of the class, phylum, etc. I prefer to use “characters-in-common” in the broader sense which would include all these characters.
Simpson (1961, ch.2) argues strongly against typological thinking in taxonomy. The point seems to be that the delineation of taxa does not entail the existence of distinguishing characters. Examples are provided by “sequences,” such as occur in many fossil series (e.g . , the classic horse line) and some neontological series (e.g., the Crustacea). Although the point is well taken, I shall proceed here as if taxa are definable by distinguishing characters, on the basis that this is usually the case. Even the Crustacea, of which it was recognized in Darwin’s time (1859:356) that members at opposite ends of the series share scarcely any distinguishing characters, are definable as mandibulate arthropods bearing two pairs of antennae, and only in some parasites and a few other forms is it known to be difficult to apply this definition.
Wherever possible, of course, groups are delineated on the basis of constellations of distinguishing characters rafher than on single characters, the risk of error in phylogenetic inference being thereby reduced. A typical example is the phylum Chordata, defined as that population of organisms, past and present, whose members possess at least in early stages of development, notochord, gill slits, and
dorsal nerve tube.
The overall workability of the group-within-group classification scheme in biology is due in part to the fact that hierarchies of constellations of distinguishing characters can so often be found on which to base the definitions of taxa. We infer from the distinguishing characters of a given taxon that it had an ancestor which possessed these characters; that there was, for example, a most recent common ancestor of chordates with notochord, gill slits, and dorsal nerve tube. This is the economical inference from the hypothesis of hereditary connection, and as such carried strong conviction. It does not entail that the characters appeared suddenly, or that they appeared together, questions usually left unanswered for lack of fossil evidence.
a. Role of Gaps
- The latter points are relevant to the question of the seeming conflict of the existence of hierarchies of constellations of distinguishing characters as the basis for the group-within-group scheme, with Simpson’s conclusions quoted above. It is to be expected from a theory of evolution by quasi-continuous transformation that, where the record of transitional forms is especially full, asynchronously graded conditions of the different characters defining a taxon should be found, making for difficulties with the definition and/or with the classification of early ancestral forms. Exactly this expectation is borne out in Simpson’s example. As he points out, the instance is exceptional in the fullness of its record. There is moreover no reason to doubt that this is the only important respect in which it is exceptional, and this interpretation is supported positively by the pronounced tendency of gaps in other instances to close with further investigation.
It seems fair to say that as the gaps are closed, the difficulties of classification generally are increased. There may be several explanations for the fact that definitions of hierarchical taxa are nonetheless so often feasible, but the prevalence of gaps is clearly a necessary part of any explanation under the hypothesis of quasi-continuous evolution. Gaps are indeed prevalent, however, and for good reason (Simpson 1960a, 1953:367) .The paradox is thereby explained to this extent: On account of the prevalence of gaps, ancestral forms with partial development of a taxon’s constellation of defining characters are rarely enough found that they do not as a rule render the definition useless. The occasional finds of missing links such as Archaeopteryx suggest the difficulties which a full record could make, while at the same time supporting the hypothesis that transformation was gradual, for some of the defining characters indeed occur in an intermediate condition in the linking form.
b. Role of Extinction.
The existence of constellations of defining characters presents also another problem, however. Typically the distinguishing characters of especially the higher taxa are complex, suggestive of long periods of genetic engineering under selection. This point is implicit in the gauntlet which Goldschmidt throws in the opening pages of The Material Basis of Evolution (1940:6-7):
I may challenge the adherents of the strictly ‘Darwinian view . . . to try to explain the evolution of the following features by accumulation and selection of small mutants: hair in mammals, feathers in birds, segmentation of arthropods and vertebrates, the transformation of the gill arches in phylogeny including the aortic arches, muscles, nerves, etc.; further, teeth, shells of mollusks, ectoskeletons, compound eyes, blood circulation, alternation of generations, statocysts, ambulacral system of echinoderms, pedicellaria of the same, cnidocysts, poison apparatus of snakes, whale-bone, and finally, primary chemical differences like hemoglobin vs. hemocyanin, etc. Corresponding examples from plants could be given.
The same point is also implicit in his assertion (quoted above) that “Certainly the evolutionary steps leading from a reptile to a bird should be infinitely more numerous than those leading from one kind of bird to another.” To mee’t this challenge without invoking the saltation hypothesis, it is necessary to admit not ,only the prevalence
of gaps, but also an enormous amount of extinction. This is because the common ancestor of a taxon with a battery of well developed distinguishing characters, e.g., the ancestral chordate or bird, represents the only successful descendant of what must have been a very long series in which experimentation on these characters was carried out. Whether or not there was side-branching of species, genera, etc., in the line from archosaurian reptile to bird, for instance (branching is to be expected, however), all but the one ancestral gene-exchanging unit evidently was eliminated before or relatively soon after the beginning of the adaptive radiation of the birds.
The difficulties with the taxonomy of the Mammalia (Reed 1960, van Valen 1960, Simpson 1959b, 1960b) perhaps reflect the fact that a fuller fossi1 record in that group has revealed many more of the blind side-branches on the way to the mammalian condition
than are known in the line from archosaur to bird. These admissions can be made without overstraining credence, however. Even together they are much less arbitrary and implausible than the hypothesis of systemic mutation, and they appear to be also in general accord with the facts. It should be emphasized, however, that the percentage of extinction, so to say, inferred here from the complexity of distinguishing characters, is very great, and the percentage of progeny-leavers correspondingly almost vanishingly small. To put the inference another way: The highly engineered characters of higher taxa, such as listed by Goldschmidt, may be described as “great inventions” (without implying that all important innova.tions have required long engineering before an adaptive radiation was possible). It is those lines in which great inventions are developed that tend to found the higher categories under this view, the rest, and even those which almost “have it,” falling by the wayside. Great inventions are a) rare, and b) enormously expensive of material and organisms, when achieved purely by trial and error. Accordingly, an enormous amount of extinction must have been involved in the founding of most higher taxa, on the basis of the tenets of the synthetic theory;
The factors emphasized by Simpson (e.g., 1959a) as being more or less peculiar to the origin of higher taxa, including rapid transformation, small populations, and new adaptive zones, each may have a role in the explanation of the matter at issue, in all cases or in some. So also may the factors of .preadaptation and multiple pathways (Bock 1959), general adaptation and character displacement (Brown 1958), and perhaps others. The hypothesis of vast extinction is in general accord with any of these principles based on the concept of quasi-continuous evolution, so far as I can see.
c. The extinction of phyla.
Simpson (1949:36) has inferred from the fossil record that extinction varies inversely with taxonomic rank; i.e . until at the class level few, and at the phylum level perhaps none have ever become extinct. [Recent discoveries in Canada indicate many.] This relationship suggests the possibility of a corresponding direct relationship between greatness of invention, so to say, and taxonomic rank, where “greatness” may be interpreted in terms of survival value for broad adaptation to, or control of, environment. The question is clouded, however, by the non-separation of the factors of diversity and divergence in the designation o( rank. To the extent that diversity enters into the designation of rank of extinct groups, the inverse relation between rank and percent extinction of taxa of that rank is not unexpected regardless of the factor of greatness of inventions; the deeper and wider the diversification, the higher the rank, and the greater the probability that at least some member species will leave progeny; conversely, if all eggs are in one basket (species), as it were, the chances of extinction are overwhelmingly great. But on the other hand, if divergence be the guiding criterion of rank, the survival of ail phyla, most classes, etc., could be interpreted to imply that the greater the departure from pre-existing modes of organization, the greater the chances of evolutionary success. Insofar as these departures rest on the distinguishing characters necessarily developed in the common ancestor, it would seem to follow, speaking of these characters as inventions, that the greatest inventions were developed earliest, subsequent inventions being of declining average merit, as judged by survival value for general adaptation. (Human intelligence is perhaps too recent a development to be judged on this scale.)
Some further light is thrown on the question, however, by consideration of the organizational base level from which the departure from pre-existing modes of organization arises, i.e., by taking the anagenetic viewpoint. The lower this level, the greater the possible departure achievable by a given amount of genetic engineering (given quantity of evolution, taken as rate multiplied by time). Thus the focus shifts from the inventions themselves to the substrate on which they are developed.
In other words: There seems to be no reason why early inventions in themselves should have greater average survival value for broad adaptation than should recent inventions involving a similar amount of genetic engineering. There is reason, however, to expect such early inventions to effect a greater proportionate change in the organization of the organism, if we take cognizance of anagenesis. There being good evidence for anagenesis on completely independent grounds, it is preferable to invoke this factor rather than to postulate a systematic decline in the average value of inventions in attempting to explain the [lower rate?] of extinction of phyla and the increasing proportion of extinction of taxa of descending rank.
Heretofore in this discussion, Goldschmidt has been answered in principle from the unamended synthetic viewpoint. With the question of the [lower rate?] of extinction of phyla, however, the possibility of a contribution from the anagenetic viewpoint emerges, as just explained. This point is not pursued here on account of the difficulties presented by the confusion of criteria of rank–not to mention the question whether the [lower rate?] of extinction of phyla is indeed a fact. We turn instead to a matter which more clearly and convincingly demands the anagenetic viewpoint.
8. DISTINGUISHING CHARACTERS OF HIGH RANKING TAXA.
It was stated above that Goldschmidt was not without reason in saying that the type of the phylum existed before the types of its classes, etc. It is in the sense that there existed a common ancestor with the distinguishing characters of the taxon that this is true. It is because the distinguishing characters of at least the very high ranking taxa tend to be so markediy embryonic and foundational (see below), as seen in present members, that it becomes difficult to explain the origin of such taxa in neo- Darwinian terms of accumulated slight variations, unless anagenesis be taken into account.
The explanation in terms of anagenesis is very simple in principle: the ancestral forms in which these characters first appeared were at such a low level of organization that the new characters in statu nascendi were not embryonic, but were perhaps even terminal,
and could later appear embryonic and foundational because they were’ built on or into a primitive substructure.
By.an embryonic character is meant one which appears early in development, whether or not it persists in the adult. Such characters may be said to be foundational on at least four counts, two of which are rather obvious: a) the prerequisites for the expression of such characters are relatively few and simple, and b) many other characters which appear later in development are often dependent upon them. To take a particularly stark example, the trunk in the developing tetrapod embryo is more foundationai on both
counts than the limb-bud, which is in turn more foundational than the digits, etc.
Comparative embryology .provides most of the evidence for the generalization that characters of higher taxa are more embryonic. It has been recognized since the time of von Baer that, at least among vertebrates, phylum characters generally appear before class characters, etc., in development, the characters of the species and varieties tending to be manifested last, sometimes not even until late juvenile or adult stages. Other phyla exhibit the same tendency with
greater or lesser degrees of aberration.
Among the green plants, for example, the presence or absence of true roots, stems, and leaves in the adult, characteristic of the division or subdivision Tracheophyta, can be forecast from quite early stages in sporophyte development. The class or subclass can often be forecast at about the same time from the number of cotyledons, while characters of order, family, etc., appear only later. On the whole it seems fair to attribute departures from this order of appearance of characters, albeit sometimes wide, to secondary distortions of time-order of development, i.e., to heterochrony. Many factors are known which may contribute to such distortions (cf. De Beer 1958).
On a somewhat less intuitively evident basis, characters of high ranking taxa may be said to be more foundational on a third count: c) they tend to be involved in the solution of problems of a more fundamental nature, i.e., common to all or most living organisms and not peculiar to the particular organism on account of the special way of life of its species, genus, or other lower taxon. From .the standpoint of the physiologist, the point may be put this way: the characters of higher taxa have to do more with problems of cellular or comparative physiology, those of lower taxa with ecological physiology.
The differences between higher taxa of animals, especially the phyla, are often differences of organ system for supplying tissues with cell foods and oxygen, and for divesting them of nitrogenous wastes and carbon dioxide, etc., while such differences rarely occur between lower ranking taxa. The biochemical differences in
types of, chlorophyll and polysaccharide which are among the distinguishing characters.of most plant divisions are perhaps even more foundational on this count, as well as on counts a) and b).
It is of course not meant to imply that evolution progresses smoothly and regularly from the solution of more fundamental to that of less fundamental problems. Populations of organisms are assumed to evolve opportunistically, so that an established mechanism is always subject to modification or even discard, with or without replacement as new situations arise. In the limpet Patella, for instance, the age-old respiratory organs of the molluscs,
the ctenidia, are missing and appear to be effectively replaced by pallial gills arising near the mantle margin which facilitate aerial respiration in the upper intertidal zone occupied by these forms. An analogous development occurs as a subclass character of pulmonate molluscs developed presumably at an earlier time; here the ctenidium is lost, and the vascularized wall of the mantle cavity acts as a lung surface in aerial respiration. The profound alteration of kidney function in the giant clam Tridacna, associated with its symbiotic habit analogous to that of the fungus component of the lichen, provides another example.
Due to the frequent functional as well as architectural dependence of less foundational characters on more founda-
tional ones, however, the latter tend on the whole to remain as a relatively constant functional and architectural substrate, characterizing the higher taxa, while the former tend to become the distinguishing characters of new species building upon this substrate.
Related to c) is a fourth count in which characters of high taxa may be said to be more foundational than those of lower taxa:
d) they tend to be more broadly adaptive (cf. Simpson et al. 1957:435ff.) On all counts, the concepts of “embryonic” and “foundational” are definable on the basis of development, without reference to evolution, although they have a rather obvious evolutionary interpretation. For present purposes, it is preferably in sense a) that foundationality should be appraised, since in this sense it is much less variable with developmental stage than when appraised in sense b). A better perspective on c) and d) is gained from physical considerations, which are deferred to part II.
9 . THE ROLE OF ANAGENESIS.
The counts on which distinguishing characters of high
taxa are more foundational than those 0f lower taxa are all explicable from the anagenetic viewpoint, and in large part not otherwise easily understood in terms of quasi-continuous evolution. The explanation, to a first approximation, is straightforward: The common ancestors of high taxa tended to be relatively simple forms, structurally and functionally; the problems which it was theirs to solve were correspondingly elementary, and the distinguishing characters developed in their solution therefore foundational in sense c), and to a degree, d). Being built on or into a simple sub-structure, they were also foundational in sense a). Subsequent characters being superposed on them, they have tended also to become foundational in sense b), i.e., embryonic.
Since the hypothesis that evolution is quasi-continuous is retained, the anagenetic viewpoint posits no greater divergence between the ancestral species from which there have been descended taxa now regarded as separate phyla, than between taxonomically adjacent recent species. But since it is supposed that the ancestral species were less complex to begin with, it follows that, as a rule, the potential divergence of their descendant taxa was greater. Thus it is implied, for example, that the insects as a group will never catch up in taxonomic rank with the echinoderm-chordate superphylum, no matter how much greater their diversity becomes, because the insects started from a much more highly organized ancestor.
This is, in essence, the answer to Goldschmidt’s taxonomic argument from the anagenetic viewpoint. It is, I submit, much like what Simpson or Rensch might have been led to had they taken into detailed consideration the problem of the origin of phyla and kingdoms, where the differences in foundationality of distinguishing characters are much more significant than at the levels within the vertebrate sub- phylum with which they have been most closely concerned. Thus, while it is true that the much lower level of organi- zation of common ancestors of kingdoms and phyla is generally
taken for granted, the macroevolutionary implications of this point have not been given their due.
As I hope to show in following sections, there are further consequences of the anagenetic viewpoint which have been also somewhat neglected, because of emphasis on other factors in recent evolutionary work.
10 . A DIAGRAMMATIC REPRESENTATION OF THE ANAGENETIC VIEWPOINT.

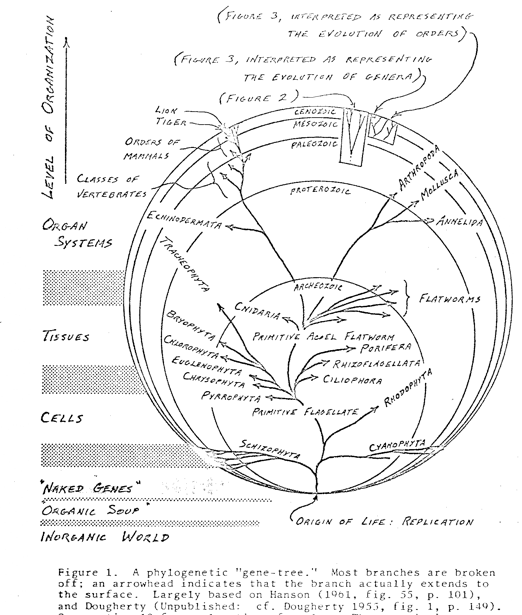
This section is devoted primarily to the explanation of a diagram, from which can be visualized some features of the preceding discussion, and which aids in making certain additional points. The diagram (Figure 1) is a modification of the familiar evolutionary tree, rather drastically pruned for purposes of argument and simplification, Figure 1. A phylogenetic “gene-tree.” Most branches are broken off; an arrowhead indicates that the hranch actually extends to the surface. Largely hased on Hanson (1961, fig. 55, p. 101), and Dougherty (unpubl’ished: cf. Dougherty 1955, fig. 1, p. 149). See section 10 for explanation of scales. The genetic interpretation is discussed in section 11.
The tree is plotted as if it grew within a balloon being blown up from the bottom, at a point which may be taken to represent the origin of life. The hypothesis of a monophyletic origin of life is not entailed, although the figure is perhaps not optimal for the representation of the ‘alternative hypothesis of a polyphyletic origin, since the same point must represent all non-abortive origins if the whole biota is to be shown in a single tree. The circles are loci of equal time; any point on the inmost drawn circle, for instance, is at the end of the Archaeozoic, except the point of origin, which is indeterminate as to time. (The peculiarities of the point of origin do not present important difficulties, at least in the context of this work.)
Level of organization is plotted vertically, on what is usually the time axis. The scale has been gauged so that the lower branch points are at a level inferred to be that of the organization of the ancestors of the corresponding high ranking taxa. Some levels are given, in common use though nowhere quantified; the hatching between them is meant to indicate that there are no sharp lines of demarcation, but rather that the levels intergrade. Not all recognized levels are included. Although evidence of the phylogeny of organisms is quite detailed in some lines, strict accuracy has not been pursued as far as possible in plotting Figure 1. This is because detailed evidence fades at the high taxonomic levels of most concern here. The limitations on evidence at these levels may be considered first with respect to depth of the branchings, and second with respect to their topological relationships (the usual concern of phylogenetics).
As to the first consideration, the ages of taxa above class level are virtually unknown, primarily because of the decline of the known fossil record below the Cambrian. Thus there is little or no direct evidence on which to base the plotting of depth of branch points of the phyla and higher taxa. They have therefore been plotted as to depth in part on the basis of a generalization drawn by Rensch from data on known ages of animal taxa, to the effect that the age of each of the major categories, species, genus, family, order, and class, is very roughly double that of the next lower category (see Figure 2). This generalization has been extrapolated to categories above the class level, so that phyla are taken to be on the order of twice the age of classes, kingdoms twice the age of phyla, but allowing wide deviation from this rule.
The above inference of a “quasi-geometric” increase of ages of categories is conditioned by the existence of a degree of arbitrariness and instability in taxonomic ranking; by variations of criteria of rank in different major groups, worked by taxonomists with different traditions; by differences of the evolutionary process in different lines, e.g., where neoteny is involved, and across adaptive zones, e.g., faster rates on land than in sea; and by uncertainties as to strata of origin and as to ages of these strata.
The inference is accordingly very inexact; yet, in favor of its general plausibility, the average age differences are so great as apparently to outweigh the uncertainties of dating, etc., in categories from sub-species to class where data are obtainable. Extrapolation beyond the class level involves, to be sure, still more uncertainty, but may be justified as the only existing rational basis for estimating depths of branch points at higher taxonomic levels.
The age-doubling phenomenon may find a measure of explanation in terms of the criteria of rank discussed in section 4. Taking divergence as the dominant criterion, the degree of divergence between adjacent taxa implicitly required for their ranking at a given level may vary quite non-linearly with level. Suppose, for example, that a minimum “index of divergence” of 0.01 is generally required for classification into different species (neglecting other possible requirements). If the implicit minimum indices for classification into different genera, families, orders, and classes were then respectively 0.05, 0.10, 0.20, and 0.40, and if the evolutionary rate of accumulation of new characters and other divergences were essentially constant, it would be expected that the average ages of categories show the kind of quasi-geometric relationship actually found by Rensch.
A complete explanation of the age-doubling phenomenon may involve also the consideration of extinction patterns. If Darwin’s idealization (Figure 3) of the process of diversification under natural selection be taken to represent the expected pattern, it is evident that the actual pattern, idealized in Figure 2, is somewhat unexpected. The implications of this discrepancy are not completely clear to me, but they suggest at least that extinction is a factor deserving of reconsideration in the pursuit of an explanation of the facts of macroevolution.
The approximate doubling procedure places the origins of the phyla of higher animals in the upper third of the diagram, i.e., no earlier than the Proterozoic. Thus they began at an organizational level nearer to that of their highest modern representatives than to that of the first living forms, assuming that the scale of levels of organization is not unduly expanded at the base. This implication of the diagram is not unwarranted in the light of their distinguishing characters (see below).
There is a degree of further substantiation of the inference of the relatively late origin of the phyla, and/or an indication that the scale of levels of organization is not unduly expanded at the base, in the well-recognized complexity of the cell. It is commonly supposed that the evolution of the nucleated cell took perhaps as long as all the presumably subsequent evolution of multicellular forms (other than blue-green algae); this supposition reflects the biologist’s high respect for the organization of the cell. If only half of all evolutionary time has been available for the evolution of the eukaryate kingdoms, however they be defined, so much the less has been available for the evolution of most of the phyla.
Concerning topological accuracy, the relationships of the branches to one another are of course inferred from other data, notably those of comparative anatomy and embryology. An attempt has been made to reflect the consensus of phylogenists in plotting these relationships, but no pretense is made that the figure portrays positively known facts. The correct interconnections of the deepest branches are not much better known than are the correct depths of the branch points, despite the much greater concern with topological relationships from Haecke1 to the present day; a review of the uncertainties is provided by Kerkut (1960)..
By drawing the tree in this way, a group which has remained at a low level of organization, such as the bacteria, can be pulled off, as it were, and made to creep along near the bottom indefinitely, while a rapidly advancing line is shot vertically towards the top, and a group advancing at an intermediate rate is angled off appropriately. Changes of rate of advance can be indicated by changes of direction up or down. As the tree grows, some of the higher forms of life, represented by upper branches, reach levels of organization not previously existent in nature. Social organization may be thought of as included in the figure, to the extent that it involves interactions within but not between species.
Although the diagram represents the anagenetic evolution of organisms, it deserves mention that, in .principle, a similar figure might be drawn for the evolution of ecosystems. The origin of life may be taken to involve also the origin of ecosystems, which, like their constituent organisms, have tended to progress upward from a lowly and poorly organized condition. While marginal ecosystems such as those of very high altitudes and latitudes may have progressed relatively little for very long periods, some of the most complex have continued to bring to realization ever higher organizational phenomena. There has been presumably an evolutionary continuity of ecosystems, with branching and rebranching as in the phylogenetic continuity of organisms. In the case of ecosystems, however, there can be little doubt that the picture is complicated by anastomoses to a much greater .degree than in the case of organisms. Moreover, the facts of branching and anastomosis of ecosystems are so little known that an almost purely hypothetical figure is all that might be drawn.
a. Discussion. To visualize the increasing foundationality of
distinguishing characters, one may consider the differences between lion and tiger, as an example of two species of the same genus, in contrast to the differences between lion and starfish, which are forms representing two different phyla of the same superphylum. There is in fact scarcely any morphological link between the latter forms except through the acorn worms, which reveal point-for-point larval similarities with some echinoderms on the one hand, and pharyngea1 gill slit similarities with chordates on the other hand. Yet, postulating archallactic departures from the ancestral course of development in chordates, it is not unreasonable to regard the differences between lion and starfish as consisting largely of accumulated slight varia- tions superposed in different ways on dipleurula-like ancestral forms.
The result is a divergence in virtually all organ systems, implying differences in paths taken toward the improvement of such basic functions as respiratory gas exchange, excretion, and sensory and response mechanisms. These are foundational differences in all senses considered in section 8. The differences between lion and tiger are strikingly superficial in contrast, and in comparing the lion with forms in adjacent genera, families, orders, etc., one may observe that the differences tend to become less superficial and more foundational. Exceptions and irregularities are to be expected in any such sequence of comparisons, but from a statistical standpoint the trend is reasonably clear. The discussion in Simpson et al. (1957:435ff.), centered on the increasing breadth of adaptation of characters of the woodpecker, may be taken as a case in point.
Aspects of foundationa1ity other than breadth of adaptation are implicit in the sequence of characters considered there. It deserves emphasis that differences consideraby more foundational than those between echinoderms and chordates occur between taxa above superphylum in rank. Often these can be related to the direction taken in mode of solution of the primary problem of living matter, that of obtaining food energy. The same may be said of many, if not most, distinguishing characters at all taxonomic levels, but at the very high levels the directions taken typically represent very basic and widely divergent possibilities. These are open primarily to living matter at a low level of organization, with little investment, so to say, in organ systems adaptive for one mode but not the other.
The sponges, for example, differ from chordates not only in respect to virtually all organ systems, as do the echinoderms, but also, to a greater extent than echinoderms, in respect to cellular and tissue differentiation. In lacking cells and tissues of exceptional irritability and contractility (nerve and muscle)., they are virtually as divergent from chordates as are the plants, and primary differences of cell differentiation of this nature are even more foundational than the differences in organ systems between the phyla and superphyla of higher animals. A partial.interpretation in the instance of the sponges is that, in their maintenance throughout evolution of
intracellular ingestion without the introduction of a single relatively large intake orifice (mouth), the ingestion of macroscopic food has never become an exploitable possibility; hence, there has been no occasion for the development of nerve and muscle tissue as adaptive advantages in food hunting. Other factors wou1d be expected to enter into a full interpretation, but it is unlikely that this one,
involving micro- and macrophagous heterotrophy as alternatives, has not played a major role. In like manner, the differences between, say, the “kingdoms” of Metazoa on the one hand and the green plants, including Chlorophyta, on the other, relate to holozoic and holophytic modes of nutrition as alternatives adopted by their respective ancestors. ·By consensus, it is supposed that these ancestors were flagellated eukaryate cells, and in forms at this relatively low level of organization, the two modes in question represented more readily exploitable alternatives than they do to complex modern descendant forms.
As implied in section 8, it is unsafe to infer that no higher metazoan can give rise to an autotrophic taxon, and no higher plant to a heterotrophic taxon. It is entirely reasonable to infer, however, that with the increased investment in organ systems adapted to heterotrophy in the evolution of metazoans, and to autotrophy in the evolution of green plants, the probability of such events has declined.
Insectivorous and hemiparasitic plants represent only partial approaches to heterotrophy; metazoans are not known to have approached autotrophy independently, although a number of lines have successfully exploited symbiosis with algae incorporated into their tissues. A return to complete heterotrophy by an autotroph is not unknown (cf. the Indian pipe and the dodder), but such events have relatively rarely been successful.
Among protists, including monerans, shifts from autotrophy to heterotrophy are much less unlikely in principle, and presumably have happened many times. In fact, simple protists, e.g., some euglenoids, are known to exhibit either mode under appropriate conditions. The opposite shift is enzymatically somewhat more difficult and is not considered to have occurred so frequently. Either way, however, the forms in question have retained a low level of organization.
Thus, among the more complex animals and plants, with relatively rare exceptions any two forms in the same genus, family, order, or class, share the same principal cell types and organ systems, reflecting retention of the nutritional mode adopted by the ancestor of the class; and to a considerable degree the same is true even at the phylum level.
The foundationality of differences across classes is not negligible, but is clearly less than that across phyla; compare respiration and circulation in mammal and fish, for example, with chordate-echinoderm divergences. The same may be said of some characters of orders, e.g., the rumen and associated structural changes (actually sub-ordinal) of Ruminantia. Across families, differences of a comparable degree of foundationality are rare; characters of cats and bears, for instance, are not all physiologically trivial (note the extraordinary specializations for preying-carnivory in the cats, and for dormancy in the bears), but even as differences across superfamilies, they manifest no major alterations of the organs and organ systems.
With respect to morphological divergences within and between genera, differences of color, or of size and proportion attributable to allometric growth are the rule, .as illustrated by cat and lion, or cat and hyaena. Rensch (1959) indeed has emphasized the role of allometry at taxonomic levels at least as high as order, as a major part of his argument in support of the hypothesis of quasi-continuous evolution in transspecific evolution. His view is compatible with the point under emphasis here, as to the greater foundationality of characters of higher taxa, but in not
considering the relative levels of ·organization of ancestors he has bypassed this point.
The examples illustrate that it is not sufficient for the understanding of a profound difference to consider only the much longer time available for the accumulation of slight differences: the low level of organization of the ancestor must also be taken into account. Only by means of extraordinarily extensive neoteny or similar mechanisms of reduction, could there be expected to appear between descendants of adjacent species of higher forms of modern
life, plant or animal, manifold differences of cellular specialization, organ systems, and body architecture of such foundationality as now exist between chordate and echinoderm, for example. That neotenic reductions and reradiations have occurred need not and perhaps cannot be denied, but such factors cannot be invoked as the general explanation for the greater foundationality of characters of higher taxa. Anagenesis alone offers an explanation, whether or not neoteny
is an important factor. Thus the same argument can be brought against general explanation by neoteny as by saltation, although additional arguments against the saltation hypothesis do not apply to neoteny. It follows from this argument that if new phyla and . kingdoms are to arise in the biota, they will most likely arise from lowly organized forms.
It is implicit in the foregoing that, as a rule, the more foundational a character, the higher the rank of the taxon which it distinguishes, and the lower the level of organization of the ancestor from which the taxon is descended. This may be regarded as essentially another summary statement of the anagenetic viewpoint. Allowing for exceptions, several classes of facts, notably from embryology, appear to be in accord with this generalization, at least at the high taxonomic levels of primary concern here, if not at all levels (see section 8).
It may be objected, however, that the generalization is drawn by circular reasoning–that taxa are ranked high because their defining characfers are regarded as foundational. This objection merits some discussion. Were diversity the chief criterion of rank, the objection would fall for the reason that commonness of a distinguishing
character would then be the primary measure of the rank of the taxon which it defines, and any correlation with foundationality would be strictly secondary. As explained in section 4, however, divergence outweighs diversity in the determination of rank, and the objection then turns on the question, to what extent is foundationality of distinguishing characters a criterion of divergence?
In practice, jt is quite possible that foundationality of distinguishing characters has been used implicitly to some extent as a criterion of divergence, therefore of rank: this usage would seem to lie behind the “Bauplan” concept, for instance. In theory, however, divergence can be conceived as entirely a matter of relative numbers of characters-in-common, without regard to their foundationality; such a concept is indeed developed to a degree in section 4. From this standpoint, which is at least as defensible as one based on foundationality ab initio, a correlation which might be found between foundationality of distinguishing characters and taxonomic rank would again be secondary, therefore not a result of circular reasoning.
It would seem to require a reclassification of the higher taxa on the basis of an index of homology or its like (section 4), in order to settle the question objectively. If the result of such a reclassification were substantially the same as the present classification and were taken as a standard, it could be concluded that our generalization is not tautological. To get the necessary information might be a rather difficult and expensive undertaking, even assuming no difficulty in principle with the problem of the “dissection” of organisms and their life cycles into essentially equivalent, discrete characters. In the absence of this information, only a subjective judgment is possible.
It is difficult to make such a judgment. The taxonomist attempting to define very high taxa may tend to give much weight to characters which are foundational in one or more of the senses considered in section 8. His choice of characters, however, may be made on the basis of their stability or reliability rather than on the basis of their foundationality. The phylogenist dealing with relationships between very high taxa implicitly accepts the anagenetic concept of evolution. His prime concern, nevertheless, is with evidence of continuity. Stable characters of great breadth of distribution are of primary importance in this concern, without particular regard to their foundationality. But if stability and breadth of distribution are primary criteria, once again the correlation with foundationality is a secondary matter, and the generalization, that the higher the taxon the more foundational its distinguishing characters, is an independent inference rather than a tautology.
This generalization is of interest in relation to the anagenetic implications of the taxonomic system. It has been recognized since Darwin that the group-within-group character of the “natural” system of classification points to an evolutionary interpretation. Perhaps it is implicitly accepted that the system points more specifically to an
anagenetic evolutionary interpretation, but it does not appear to have been argued why this should be so. A possible reason appears if one imagines being presented with the task of forming a set of objects uniquely classifiable into a group-within-group scheme, rather than classifying a set of objects already formed. It is difficult to conceive a way to approach this problem which would not be anagenetic, at least in .the sense that. the objects formed would become increasingly highly differentiated with each lower subgroup constructed. The whole task would be made easier if the objects could be formed so as to have a development analogous to that of higher plants and animals. In that case, the natural way to carry out the project would be anagenetic not only in the sense of increasing differentiation, but also in that the attributes assigned to the highest groups would be the most foundational, at least with respect to being “embryonic” and being built upon by attributes subsequently assigned to lower groupings; etc.
Thus, the group-within-group feature of classification alone suggests an anagenetic interpretation, and the generalization as to the greater foundationality of characters of higher taxa, insofar as valid, confirms such an interpretation.
Anagenesis is almost universally recognized as a factor in evolution, as previously noted. It is therefore perhaps inadvisable to labor the point that aspects of taxonomy also imply anagenesis. Such implications appear not to have been considered up to now, perhaps for the following reason. It is, to begin with, only in groups where considerable anagenesis has occurred, i.e., only in the higher plants and animals, that pronounced anagenetic implications of taxonomy might be expected to be shown. Within these groups the effect is not generally pronounced even at the class level, because the ancestors of classes were already forms at an organ system level of organization, judging from their distinguishing characters. In some cases it may be inferred that even the ancestors of phyla were not enough simpler than their modern descendants to necessitate con- sideration of anagenesis: the hypothetical ancestral chordate, for instance, was presumably already an animal with dorsal hollow nerve cord, notochord, and pharyngeal gill slits, as special characters ancillary to previously established nervous, muscular, and digestive systems, etc.; even its most advanced descendants can be regarded as manifesting only moderately foundational subsequently developed characters.
In short, it appears to be necessary to look back extremely far if the anagenetic implications of taxonomy are not to escape attention. Even systematic paleontology need not deal with them, if only on account of the virtual cut-off of the fossil record at the Cambrian. They do not force themselves upon investigations of the descent of phyla, etc., because the relative scarcity of data largely confines these investigations to the search for possible relationships on the basis of a few characters, and problems of formal taxonomy remain in the background.
The point is illustrated in Figure 1. Although of course the time scale is different along different rays from the starting point (origin of life), distance varies linearly with time on each ray, as on the usual time axis. The origin of life is taken to have occurred some 2.6 billion years ago.This is on the late side according to at least one recent estimate (Holmes 1954), but the choice was made with a view
to allowing as much space as possible for evolution since the Paleozoic, while yet maintaining a fair representation of the whole span. Even with this possible exaggeration of later stages, the orders of mammals appear to have arisen only yesterday, as it were. If, to a reasonable degree, time elapsed reflects anagenesis accrued, it is therefore not surprising that the existence of marked differences in foundationality of ordinal as against genus and species characters has not been urged. The origins of vertebrate classes are also relatively close to the surface, and the same conclusion is accordingly illustrated to a lesser degree at this taxonomic level.
In phyla other than Chordata the orders, etc., tend to be older, but since the average ages of even the classes are calculated not to exceed 540 million years (Rensch 1959:92), the general conclusion still is reflected in the figure, that orders and classes, if not also many phyla, arose a relatively short time ago from relatively highly organized ancestors, compared to the time since the origin of life and
the presumed molecular level of organization of the first living forms.
It has been indicated above that Figure I has been constructed in part by means of rough extrapolation of Rensch’s diagram (Figure 2). Conversely, Figure 2 can be viewed as a small section of the more general figure, as shown in the inset. This view reveals how shallow a portion of the entire evolutionary picture was diagrammed by Rensch, hence suggests one reason why he could have been unconcerned with the anagenetic viewpoint of the origin of higher taxa. Likewise, the lone figure in the Origin of Species, which was introduced by Darwin for visual aid in the description of the process of formation of new groups (diversification, adaptive radiation, or cladogenesis in modern terms) appears as but a small part of Figure 1. Darwin’s main explanation was of the formation of genera, and on this basis his figure is represented by the very small corner inset in the general figure–the inset within the inset.
It is interesting that Darwin was clearly of the conviction that
the same figure portrayed the formation of higher taxa, but that he did not systematically carry it beyond the order level, i.e., beyond the point where his figure is represented by the larger inset in Figure 1. Thus he says (1859:IV,108-9):
In the diagram, each horizontal line has hitherto been supposed to ·represent a thousand generations, but each may represent a million or a hundred million generations, and likewise a section of the successive strata of the earth’s crust including extinct remains. I see no reason to limit the process of modification, as now explained, to the formation of genera alone. If, in our diagram, we suppose the amount of change represented by each successive group of diverging dotted lines to be· very great . . . (there will be formed) two distinct families, or even orders, according to the amount of divergent modification supposed to be represented in the diagram.
In regard to further extension, he says in the last, recapitulatory chapter (1859:XIV,409):
It may .be asked how far I extend the doctrine of the modification of species. The question is difficult to answer, because the more distinct the forms are which we may consider, by so much the arguments fall away in force. But some arguments of the greatest weight extend very far. All the members of whole classes can be connected together by chains of affinities, and all can be classified on the same principle in groups subordinate to groups. Fossil remains sometimes tend to fill up very wide intervals between existing orders. Organs in a rudimentary condition plainly show that an early progenitor had the organ in a fully developed state; and this in some instances necessarily implies an enormous amount of modification of descendants. Throughout whole classes various structures are formed on the same pattern, and at an embryonic age the species closely resemble each other. Therefore I cannot doubt that the theory of descent with modification embraces all the members of the same class. I believe that animals have descended from at most only four or five progenitors, and plants from an equal or lesser number.
Analogy would lead me one step further, namely, to the belief that all animals and plants have descended from some one prototype. But analogy may be a deceitful guide. Nevertheless all living things have much in common, in their chemical composition, their germinal vesicles, their cellular structure, and their laws of growth and reproduction. We see this even in so trifling a circumstance as that the same poison often simiiarly affects plants and animals; or that the poison secreted by the gall-fly produces monstrous growths on the wild rose or oak-tree. Therefore I should infer from analogy that probably all the organic beings which have ever lived on this earth have descended from some one primordial form, into which life was first breathed.
It is clear that Darwin had the intuition of the full generalization of his diagram to the representation of the whole tree of life. It would appear, however, that as a result of his concentration on the origin of species, where facts were and are least remote and most abundant, the difficulty presented by the greater foundationality of differences between the very high ranking taxa escaped his attention. In effect, therefore, Darwin worked in too shallow a layer of Figure I for differences in levels of organization of ancestors to make their effects strongly felt. One may speculate that the macroevolution controversy might have had a very different history, had Darwin systematically pursued the analysis of his figure to deeper levels.
11 . THE GENETICS OF ANAGENESIS.
Although little Or nothing is known about the genetics of anagenesis, it is worth while to consider the subject because of its intrinsic importance, and because of the implications for it suggested by the anagenetic viewpoint of macroevolution.The general line of thought in this section is as follows. Anagenesis evidently entails the accumulati?n of genetic information by living matter, whether considered from the standpoint of organisms or of ecosystems. From the former standpoint, the prevalence of recapitulation of evolutionary stages by developmental stages of the organism suggests that, subject to many exceptions, new genetic information has to do with terminal stages of development, building on the information already in store. Since conscious thoughts and actions can be regarded as being among the terminal phenomena of development, it would seem to follow that new genetic information is involved in the determination of these thoughts and actions. At this level, however, there are limitations on the possibility of verification of the hypothesis, more serious than mere limitations of technique. Alternative hypotheses therefore cannot be excluded, and their validity might mean that genetic information plays a more passive role in.the determination of phenomena of life than is implied by the “DNA dogma.”
In the words of an anonymous geneticist, “Life is DNA’s way of making more DNA.” This concisely expresses the guiding principle of modern molecular biology, which seeks to interpret all phenomena of life in terms of chemistry, hence of physics, in a comprehensive naturai philosophy: The last two decades have yielded results giving great impetus to this approach (cf. Sinsheimer 1: 57); yet its strength is perhaps best indicated by much older considerations. The anonymous geneticist was anticipated by August Weismann with his classic idea of the continuity of the germ plasm, according to which the organism is merely the temporary vehicle for germinal material of the sex cells. It may be argued even that the essential “facts of life” have provided a basis since the dawn of thought for such a viewpoint, wherein the germinal material, for all its minuteness, emerges as the only truly valuable substance in nature, it alone being alive since time immemorial and-potentially immortal, all other values being dependent upon its existence. Aspects of the division of labor among the various species of the biota were also visible to the ancients, although a broad view based on recognition of the photosynthetic role of plants seems not to have arisen before the nineteenth century.
It is only recently, in ariy case, that there has developed a view of life as the collective activities of the gene pool of the biota, in which the energy transformations (catalyses) of the individual units of genetic information are seen as coordinated in the successively higher integrative levels of organism, society, ecosystem, and ecosphere, to produce an approximately balanced evolving thermodynamic system, somewhat analogous to an engine, operating between sunlight input and earth’s heat output energies; It “is not yet clear whether a fully satisfactory general theory of biology can be worked out on these reductionist lines (cf. Bray 1958, Elsasser 1961). It is not even undisputable that DNA is the macromolecular basis of life (cf. Kavanau 1960). It is clear, however, that genetics, as the science of the replicating molecules which are the transmitters. of hereditary potentialities, is fundamental to biology in general and to evolution theory in particular.
The anagenetic viewpoint implies an accretion of information in the genomes of organisms, for which, hoever, little or no direct evidence has been as yet obtained. Accretion is an obvious necessity from the overall view, comparing those forms (“naked genes”) which could arise with reasonable physico-chemical probability as the first living things, with modern forms. It follows that a major chapter of evolutionary genetics should have to do with this accretion. In fact, however, evolutionary genetics in studying the spread of alleles under pressures of mutation, selection, and drift has tended to skirt this problem. It is as if evolutionary genetics postulated genomes composed of decks of cards of constant number, and concerned itself with the establishment or elimination of mutational alterations of the “fine structure” of these cards through shuffling and selection. From the anagenetic standpoint, on the other hand, the growth in the number of cards in the decks is of central evolutionary interest.
To be sure, this probiem has not gone unattended (cf. Weir 1946, Lewis 1951, Stephens 1951, Goldschmidt 1955; the topic is briefly reviewed by Dobzhansky (1951:294ff.), but it appears to be
more than experimental difficulty which is responsible for its being given a minor place in evolutionary discussions.
Even those writing on the problem of evolutionary innovation and the appearance of new structures have not dwelt on the accretional implications of innovations in their totality. The emphasis has been, rather, on the conservative aspects of innovation, from which standpoint the role of genetic accretion is naturally minimized. Thus Rensch (1959) stresses the degiee to which allometric growth is involved in evolutionary modifications of all sorts, and therewith
stresses quantitative as against qualitative factors, presumably interpretable in terms of alterations (mutations) of genes already present, which affect rates of activity of these genes. He says (p.290):
This princip1e–the origin of new organs as a consequence of a strictly quantitative increase of certain tissues causing new growth gradients–is applicable to many animal types and has been too much neglected in evolutionary thought. It is especially important to notice that such an increase of a tissue may not serve any special function at first. Later on, however, the “superfluous” tissue may be “employed” by a new function in the course of subsequent evolution.
Mayr (1960), in a still brqader and generally excellent review of the subject of evolutionary innovation, likewise emphasizes the degree to which it can be understood in terms of modification of what was already present, notably through intensification of function and change of function. In a brief consideration of genetic aspects of the problem, Mayr makes no mention of the question of accretion of genetic information.
The gradual and conservative nature of most, if not all evolutionary novelty, is interesting and important especially with regard to discrediting the saltation hypothesis. It is indeed this motive which no doubt largely underlies the emphasis of these authors on gradualism and conservatism, which emphasis was not undue. There appears to be no necessity to depart from any of the principle conclusions drawn from this approach, but the anagenetic viewpoint brings the problem of accretion of genetic information out to the center of the stage, whereas the unamended synthetic viewpoint, if it may be put so, represented by Rensch and Mayr, leave this problem in the background to the extent that one might infer that it is not important.
As already indicated, it is another matter to say anything constructive about the problem. The stage is perhaps only beginning to be set for its formulation and investigation, insofar as the setting involves the synthesis of Darwinian and Mendelian ideas not achieved until the 1930′s, and the still later, fusion of genetic and biochemical ideas of the 1940′s. The following discussion may serve as a useful base of reference for approaching the problem, albeit subject to serious criticism.
a. An idealization: the “gene tree.” By making certain assumptions, the “tree of life” may be looked upon as a tree of DNA or of genes rather than of organisms. Such a genetic interpretation of the tree of life is on the one hand further removed from the realities of fossils and comparative anatomy, etc., than is the usual interpretation, but is on the other hand closer to fundamentals from the standpoint of molecular biology. The “gene tree” is presented as perhaps the simplest possible genetic representation of evolution as a whole, exclusive of population phenomena, which could be expected to bear a reasonable degree of correspondence to reality.
The necessary assumptions reflect most of what is meant by “signal” in this work. They are as follows:
i. An organism’s genome(s) adequately represents the organism itself, i.e., there is information encoded on the genome corresponding to each of the organism’s charaaters and potentialities. Some possible limitations on this assumption are discussed in part c. of this section, but there is relatively little question of its general applicability.
ii. The genetic determinants of characters a·nd potentialities act independently of one another. There is no doubt that this assumption is not strictly valid. It would be hopelessly invalid if position effect played the generalized role postulated by Goldschmidt (1955), involving hierarchies of fields of action, corresponding to hierarchies of blocks of genetic material ranging in size from the entire genome perhaps to individual nucleotides. Position effect is, to be sure, a well-known phenomenon, but the usually minor phenotypic effects of viable inversions, translocations, etc., even in accumulation, argue against Goldschmidt’s view of their possible overriding importance. Figuratively speaking, it appears that the order of letters, words, and perhaps sentences is important in the coordination of the genome’s message, as well as important to its meaning, but that the order of paragraphs and larger units makes relatively little difference. Thus developmental order, for example, appears not to depend in an important way on visible chromosomal order, although it probably depends greatly on nucleotide order in small lengths which constitute genes.
iii. An assumption corollary to the second is that an organism’s genome can be represented by a single line or chain, with the genetic information arranged in order of age, from most ancient to most recently incorporated. No connection of this linear arrangement with chromosomal order is implied, although such a connection-may exist within limits, as discussed in part c. of this section. A difficulty may appear to exist if the well-known compression of developmental
stages, as new terminal stages are added in phylogeny, entails that old genetic material is freed and made available for terminal acquitions. It might be answered that the genetic information no longer used is perhaps suppressed rather than freed, but the greater likelihood is that both suppression and freeing occur. In any case, the difficulty is largely removed if it be granted that the recoding of the old genetic material results in essentially new genetic information.
iv. Possibly more problematical is the assumption that homologous characters of related organisms–structural, functional, behaviora1–ref1ect homologous genetic material and information. In the sense that genes of homologous characters are descended from common ancestral genes there can be no quarrel with this assumption, given the homology of the characters; but it must be admitted that where two species or higher taxa have retained a trait in common, the genetic determinants of the trait may have shifted kaleidoscopically in very different ways during the descent of the two taxa from a common ancestor, even to the point of altering the order of nuc1eotides so that little or no common information could be detected were we to know this order. Although Dobzhansky (1959) appears to expect such shifting to have been the rule, such .facts as the correspondence of gene order in different species of bacteria, the similarities of insulin molecules in distantly related mammals and of hemoglobin molecules in vertebrates, the point for point correspondence of bands in segments of the salivary chromosomes of different species of Drosophila, and the phenomena of vestigial organs, lend credence to the opposite assumption (see Emerson 1961).
It is obvious in any case that the assumption loses plausibility as we go to homologies at higher taxonomic levels, but there remains something in its favor at even the highest levels, in virtue of biochemical ,homologies at these levels, suggestive of genetic “blueprints” in common by descent.
v. The fifth assumption is one whose exceptions are a matter of record rather than of hypothesis: the line-up of genetic information by age corresponds to a line-up by time of action in development. The exceptions involve departures from perfect recapitulation, as discussed later in this section.
There is no question that, in a representation incorporating age and time of action into the line-up of genetic information, ,a branching and rebranching arrangement, perhaps with anastomoses, would allow for a better correspondence with reality than would a strictly linear arrangement; heterochonies and even some pleiotropies could then be explained in principle. A fully adeciuate representation would doubtless also involve feedbacks and other complications, particularly to allow for the effects of interaction with a variable environment. Yet, the simple linear arrangement presented here permits the representation of a considerable body of “signal” phenomena, and is useful as a first approximation to reality, provided that the nature of its limitations be not forgotten.
If the genome lines of two different organisms be compared, it would be found, according to our assumptions, that a proportion of the lines could be synapsed, corresponding to the proportion of characters-in-common, or to the index of homology. The index developed on the idea of the analysis of morphological and other characters thus lends itself to a genetic interpretation, as previously noted (section 4). If recapitulation were exact, the synapsing proportion would include all genes from oldest up to the time of the most recent common ancestor. A phylogenetic tree such as Figure 1 would then represent a summary of such comparisons, where a given species, i.e., its genome, is represented by the line from base of tree (origin of life) to tip of branch, rather than by branch tip only.
Allowing for the synapse of alleles, individuals of the same species are all represented by essentially the same line. Closely related species will be represented by lines which synapse
everywhere except for a short length at the tip (cf. tiger and lion), while distantly related forms are represented by lines which synapse only near the base.
This scheme, in effect a genetic interpretation of Figure 1 which permits looking at the familiar tree of life as a “gene tree,” is submitted as an idealization from which probably every actual comparison would reveal some departure, and from which the departures in some cases would be perhaps enormous. It is therefore to be regarded as a generallzation with predictive value of a low order of probability at best. The idealized scheme provides a skeleton or framework, .however, on which could be based the discussion of many aspects of evolution in addition to the problem of genetic accretion. It may be taken to represent the basic “signal” of the evolutionary process, departures constututing “noise,” albeit often interesting and important noise.
According to the signal, a character is fixed once it has proved its adaptive value and has had its genetic determinants established in the gene pool of a population–in Waddington’s terms (see 1960a and earlier references given there), assimilated–and subsequent splitting of that population will be on the basis of new characters, less foundational as a rule because dependent on the pre-established characters: Thus terminal acquisition, of new genes. together with new potentialities and new characters, is implied to be the basic
signal represented by the idealization.
Since perfect recapitulation (repetition of phylogeny during ontogeny) is entailed if all genes maintain their expression in order of acquisition, the “biogenetic law” is also implied to be basic signal.
The departures from perfect recapitulation have been rather well investigated and described at an embryological level (cf. De Beer 1958). [Knowledge of these departures at a genetic level is a major feature of current DNA research.]. Since the “noise” is so great at the embryological level that the biogenetic law has fallen into widespread disfavor, and since the noise at the genetic level must be assumed to be at least as great and perhaps greater if Dobzhansky (1959) is right, there might appear to be no reasonable basis for making a schematic idealization of the evolutionary process as done here, and no advantage in it. There is at least as much evidence for signal as for noise, however, and the simplification provided by the idealized scheme might be useful and suggestive if used with caution. Its general plausibility is considerable, judging from the following incident, which reveals a critical senior student of both genetics and embryology entertaining essentially the same point of view. (I am unable to judge to what degree I may have been influenced thereby in coming to it myself. )
In 1952, C. H. Danforth began a class lecture on the topic of developmental genetics, by pointing out that embryology’s greatest generalization–that development repeats phylogeny–insofar as true implies that genes determining characters of high ranking taxa, since they act first in development, appeared first jn phylogeny, with the genes determining characters of succes&ive lower taxa arising in correspondingly later times. Professor Danforth’s immediate inference from this was, incidentally, the answer to Goldschmidt’s argument which I had recently conceived independently, and it was thus that I learned of his priority: The ancestors of higher taxa were typically simpler forms than were the ancestors of lower ranking, derived taxa, genetically as well as morphologically; hence, on account ,of the greater possibilities .of subsequent divergence, siblings of a sufficiently simple form might be ancestors of different phyla. The simplicity and obviousness of this idea, I may be permitted to add, should not on the one hand keep it from being given its proper due, and does not on the other hand make it easy to establish.
b. Recapitulation. In view of the present-day ill repute of the biogenetic law, the gene-tree’s concordance with that law makes it advisable to look further into its status.. Moreover, the anagenetic viewpoint suggests an identity in essential content of the laws of von Baer and of Haecke1, the non-recognition of this identity having perhaps contributed to the disillusionments of the first half of the 20th century (see for instance De Beer 1930 et seq., Shumway 1932, Meyer 1935, Wilson 1941, Holmes 1944, and the extensive review of facts and interpretations in Rensch 1959:239-66).
Before Darwin, recapitulation generally implied a climbing of the “Scala Naturae” by an organism during its development. Some of the stigma associated with the term may hark back to this pseudo-scientific interpretation. A degree of parallelism between embryonic stages of higher forms, adults of lower forms, and fossil series provided the evidential basis for the concept. The eminent von Baer’s examination of facts compelled him to reject it, and he advanced what is in part a contrasting concept in his principle of divergence, according to which embryos resemble each other most closely in early stages and gradually diverge, manifesting characters of the phylum, class, order, etc., in sequence during the process, this being at the same time the order of characters from most general to most speclalized: As Shumway puts it, it is sometimes forgotten that von Baer’s position was in direct opposition to the theory of parallelism. “It is an ancient scandal that Haecke1 in citing von Baer’s conclusions to support his own theory deliberately suppressed the fourth: ‘Fundamentally the embryo of a higher animal form never resembles the adult of another animal form, but only its embryo’’ (Russell trans.).’” Von Baer’s divergence principle is clearly out of keeping with the idea of a linear “Scala Naturae” of organic beings, therefore out of keeping with recapitulation in its original sense. But the “Scala Naturae” was discredited by Cuvier (see Eise1ey 1958), and once it was established that there were many independent scales of being, as it were, the principle of divergence became.much less out of keeping with recapitulation in the sense of an organism’s climbing the scale of its own group during development.
Finally, Darwin’s idea of branching evolution (not Lamarck’s primarily linear idea) made the phenomena ‘of parallelism and divergence comprehensible under a single viewpoint, that of recapitulation in the modern evolutionary sense.
It would appear that the concept as revised and championed by Haecke1 came into difficulties in part because he believed that the embryo resembled the ancestral adult, and in part because of his tendency to view the adult form of the ancestor as a cause of the corresponding developmental stage of the embryo, without suffitient regard to the intervening proximal causal agents.
Consider first the latter difficulty. With the rise of genetics and experimental biology, p6ssible explanations could be advanced in terms of mutation of genes affecting various developmental stages. Thus Shumway (1932), elaborating on Lillie’s discussion of 1908, posited terminal acquisition of new characters together with modifications of earlier characters, including reductions and shifts of relative times of appearance of the latter, thereby explaining in principle both the residuum of recapitulation and the aberrations from it, particularly the tendency for organs and organ systems to show relatively accurate recapitulation while the organism as a whole might depart .rather widely therefrom. The genetic interpretation which this suggests has been discussed above: in brief, the terminal characters could be the effects of new genes, and the earlier modifications the effects of mutation of earlier acquired genes and shifts of their times of action in development.
To many minds the aberrations, including a number of types described by subsequent students, were of such extent that the notion of a law of recapitulation was to be abandoned. Shumway (1932:98) for example, expressed the opinion that those of us who were reared in the phylogenetic tradition may see it go with a sigh of regret. Those of us charged with the’responsibi1ity of expounding the law of evolution to our classes will miss a familiar maxim, easily learned and a convenient ske1eton on which to hang the discrete data of embryology. But there can be no excuse for continuing to impress plastic minds by means of discredited generalizations. Let us rather return to the laws of von Baer and explain the resemblances which these describe in terms of processes rather than of precedents.
This opinion is subject to the following criticism, however. If the opponents of Haeckel are willing to return to the laws of von Baer, it must be because the aberrations from phylogenetic sequence during ontogeny are not sufficient to obscure the ground-base of recapitulation. If it be granted that von Baer’s generalizations have even a moderate order of validity, it is reasonable to look upon perfect recapitulation as “signal” and the departures from it as “noise,” the more so since, from the modern molecular or biochemical genetic point of view, both signal and noise are
eminently reasonable expectations. It must be inferred that even De Beer is in substantial agreement with this appraisal, in the light of his statement (1930:102) that “If only the recapitulationists would abandon the assertion that that which is repeated is the adult con- dition of the ancestor, there would be no reason to disagree with them.” It remains then to consider the second objection to Haecke1, which is also subject to criticism.
It is today a commonplace that everything now alive has had equal time to evolve, so that one· does not look to living forms for the adults which may have been recapitulated during an organism’s embryonic development, except as “living fossils” may represent adult ancestors. According to Meyer (1935), T. H. Morgan credits Agassiz, in the Lowell lectures of 1848, -with proposing “for the first time the theory that the embryo of higher forms resembles not sp much lower animals living at the present time, as those that lived in past times.” In short, the parallelism of fossil series with development is closer than that of living series. This is substantiated by phenomena such as an “Archaeopteryx” stage in the development of birds (Steiner 1938; see Goldschmidt 1940:389).
In extending this principle back beyond the fossil record, to what might be called an Age of Embryos or Larvae, a merger of the laws of Haeckel and von Baer suggests itself. Insofar as the adult ancestor was embryonic by modern standards in regard to its level of organization, it was an embryo, so to say, and both “laws” are correct. In recapitulating the embryo of its ancestor, the modern embryo also recapitulates the adult, except that many superficial characters of the ancestral “larval adult” have doubtless been shed in the modern embryo (notably protective shells, etc.), while non-terminal modifications have also occurred as noise tending to obscure the basic signal.
Thus the anagenetic viewpoint may illuminate the status of the biogenetic law by indicating an equivalence, albeit imperfect, of the principles of Haeckel and von Baer. This” is not to say that the limitations of tfle validity of the biogenetic law are reduced. It has been established that aberrations from perfect recapitulation, particularly its heterochronies, are so universal that one cannot look upon any embryo as a reliable model in respect to all organ systems, etc., of the ancestral “embryonic adult.” It is in this sense that critics of the biogenetic law are justified in regarding the great generalization as discredited. It remains true, however, that only the fossil record, where extant, outweighs comparative embryology as a source of evidence for the history of a form. The fact that inferences made from such evidence are subject to uncertainties is not a fundamental objection, since this is true of all scientific inferences, to varying degrees. Independent validations of inferences from embryology as a result of fossil findings, though not extensive and never perfect, seem to be sufficient to indicate that the degree in this instance is not too extreme. Therefore it is going too far to dismiss the biogenetic law as inadequate for a convenient “skeleton on which to hang the discrete data of embryology.” It is as such a skeleton that it is incorporated in the gene-tree, and primarily on grounds acceptable to the critics themselves, viz., the general validity of the principles of von Baer.
While the anagenetic viewpoint of the origin of higher taxa permits a natural deduction of recapitulation, the saltation viewpoint does not. Goldschmidt (1940:389) argues that:
The presence of recapitulation shows positively that the original mutational change in the ancestors affected development after the stage which is recapitulated. The fact that recapitulation is an ubiquitous feature of development suggests that macroevolution has progressed mainly by this type of change.
Thus he brings the saltation theory into line with the embryological generalization, and the high level of differentiation of most characters of high ranking taxa (see section 7) does indeed leave a loophole for saltation in their formation, though not a very plausible one on account of the intricately coordinated organization of these characters, which speaks for long cumulative trial and error in their evolution.
It is of incidental interest that Goldschmidt says in the same place that:
The genetic change is probably a permutation of some of the genetic elements controlling development, . . . and does not require the origination of new genetic determiners or determining systems.
This indicates a lack of awareness of the “new gene problem” of central interest from the anagenetic viewpoint, as argued above.
Goldschmidt says further:
On the other hand, a genetic change inv~lving a huge qualitative departure which would completely revolu- tionize the processes of development from their very initiation, would wipe out the possibility of recapitu- lation and would mean such an immense departure that it probably could rarely if ever lead to a viable product. A viable product would be a new phylum.
Although this view would seem to be in accord with th:e facts
of the number of phyla, and is not necessarily out of accord with their great average-age, it offers a less natural explanation of their origin than the anagenetic view, and is defective in not invoking the factor of anagenesis, undeniably relevant at the taxonomic level of which Goldschmidt is speaking (which to be precise is above that of phylum). Goldschmidt’s conclusion on this topic is interesting:
Recapitulation, then, is an ubiquitous fact, unavoidable because of the method of evolution by large single mutational steps affecting rates, etc., of embryonic processes occurring at a definite time, and because of the mechanism of development built upon a timed systemof serial processes, the order of which is unalterable.
It would appear that saltations are entailed by recapitulation, an inference not easy to follow. A saltationist might be expected to emphasize the “noise” rather than the “signal”with respect to recapitulation, and perhaps the fact that G6ldschmidt has done otherwise may be taken as an indication of the strength of the signal.
c. A return to the problem of new genes.
- The gene-tree idealization has impiications for the problem of gene accretion beyond what was said in the first pages of this section. These deserve brief mention despite the speculative level of the subject at the present time. As presented above, the genetic interpretation of recapitulation and its aberrations postulates new genes, which act at the end of development, together with mutational
modifications of the nature and time of action of all pre-established genes. To a considerable extent this view represents a reasonable working hypothesis. There cannot be denied, however, the possibility of accretion of new genetic information whose action is interpolated between intermediate stages, and this undoubtedly has been a more important factor in many cases than has accretion of terminally acting genes; good examples are provided by some of the hydroids (cf. Hyman 1940:635). Here it is not unreasonable to say, however, that noise has drowned out signal.
As indicated above, a further amendment is called for by the phenomena of pleiotropy, which sometimes result in quite marked phenotypic alterations throughout most of the life cycle following alteration of a single gene. Rensch (1959) has discussed these phenomena, with special reference to the mutation of genes determining hormones.
Among the comp1icating factors may be mentioned finally the existence of levels of integration above that of organism, including family, society, and ecosystem. Evolution involves increased homeostasis in successively higher integrative levels, often accompanied by decreased homeostasis at the level of the organism, as exemplified by lowered resistance to desiccation in termites of higher social organization, and by increased dependence on the trappings of civilization in man.
As Emerson (1960) has aptly put it, “The fluctuating external environment of lower levels of organization becomes the internal homeostatic milieu of higher levels.” Evidence of the genetic basis of homeostasis or other organization of such higher units is sparse at best. Analogy suggests, however, that just as the phylogenetic and correlated ontogenetic accumulation of new structures in the organism points to an overall increase in genetic information, even
though many old structures are lost or reduced, so does the appearance of new organization of the higher units point to the same thing; moreover, this accumulation of social homeostases may involve new genes to an important degree, in keeping with the mode of accumulation of morphological characters. Thus the gene-tree idealization takes the origin of new characters and new genes by terminal acquisition to be the primary pattern of accretion.
The question as to how frequently new characters originate by terminal acquisition has been discussed by Holmes(1944) and at greater length in relation to non-terminal acquisition by Rensch (1959). Suffice it to say here that it has been generally accepted since Darwin, even by many of the strongest critics of Haeckel, including Naef, Sewertzoff, and De Beer, that new characters have commonly arisen as terminal additions to ontogeny, or anabolies (see Rensch 1959:25ff.).
The question as to how frequently new genes which act terminally are acquired is another matter, about which little is known and little has been said. Holmes (1944), adding his own points to those made by others, including Goldschmidt, has implied that developmental recapitulation with terminal acquisition may be expected on statistical grounds even without addition of genetic material, and even though mutations are equally likely at any stage; they are more likely to be manifested in later stages because their effects are easier to observe, there is more differentiated structure as medium for their expression, and they are less likely to disrupt development and accordingly more likely to be viable. These points may be accepted, however, without disallowing that new genetic information may tend to act terminally as well as that new characters tend to appear terminally in development.
Holmes’ third point, moreover, distinctly implies that it is the terminal changes, including additions, which stand the best chance to become established in the genome. The inevitable result would then be a preponderance of, acquisition of new genetic information which acts terminally, assuming merely that there is indeed an accretion of information, which assumption is virtually forced upon us by the overall picture.
It suffices for purposes of the present argument to recognize acquisition of terminally acting genetic information as an important factor in genetic accretion. The greater its preponderance, the stronger the signal and the weaker the noise by the criterion represented in Figure 1. Thus the overall validity and predictive utility of the gene-tree idealization is correlated with the relative strength ofthe signal factor of terminal acquisition.
Consideration of possible mechanisms which would provide new genes with terminal action leads to philosophical issues. As briefly as possible, an attempt is made here to show how these issues arise and to indicate some of the factors bearing on them. Insofar as old, early acting genes were once new and terminal in action, the mechanisms and issues in question are of general rather than peripheral interest.
Perhaps the simplest possible mechanism of terminal acquisition in the intended sense would be that of continued tandem duplication of new genes, with corresponding tandem additions to biochemical pathways after suitable mutation of the extra genes. It is not impossible that this diagrammatically oversimple concept actually represents reality to a substantial degree; adjacent genes controlling sequential biochemical steps are known, from studies of bacterial recombination (see, e.g., Beadle 1957 for a brief summary with some references). There can be 1ittle doubt, however, that other mechanisms .play a role: among the most likely are that mentioned earlier, of the reemployment of obsolete genes and also that of the reemployment of entire extra chromosomes and genomes, resulting from aneuploidy and polyploidy (cf. Dobzhansky 1951). These supplementary mechanisms are not necessarily out of keeping with the idea of terminal action of new genetic information, but undoubtedly they do not exhaust the possibilities among effective mechanisms.
It would be of much interest to be able to relate Waddington’s “genetic assimilation” to the mechanisms of incorppration of new genetic information. The phenomena whose genetic assimilation has been studied are relative1y superficial rather than foundational. It might be argued that the new terminal characters postulated here to be the primary feature of anagenetic evolution may be former exogenous traits which have become established through genetic assimilation. If this is so, it suggests further that from the anagenetic viewpoint, even the most foundational characters may be former terminal exogenous traits which have long since been assimilated. Virtually nothing is known of the mechanisms behind the process of genetic assimilation, but it is not unreasonable to suppose”that this process involves the acquisition of new genetic information. This train of thought suggests the possibility that the ptocess of genetic assimilation has been linked with the processes of new gene acquisition throughout evolution.
Furthermore, a link between these processes and the phenomena of consciousness is suggested by the fact that conscious experience may be looked upon as being among the terminal phenomena of development. Consciousness after maturation is principally at issue here, and not necessarily all aspects thereof. The relationship applies to aspects of animal evolution, and may apply to plant or protist evolution only to a limited degree, at best.
The introduction of phenomena of consciousness into the argument is impelled by the ideas of Schrodinger (1958). From observation in personal experience of the shift of consciousness always toward new problems, with the shunting of solutions to former problems into the background of the subconscious, Schrodinger was led to posit that the same shifting from consciousness to subconsciousness may have occurred in the phylogeny of nervous processes as well as in their ontogeny; beyond this, he suggested that what is “a property of nervous processes is a property of organic processes in general, namely, to be associated with consciousness inasmuch as they are new.”
Thus, one might say, the genes contain the stored solutions to a phylogenetic series of problems which, for all we know, may have been “conscious” problems when they first arose–dimly conscious in the early history.of life, increasjpgly intensely conscious with the ‘ anagenetic evolution of nervous systems. Solutions to these problems then may have been gradually transformed into “unconscious” mechanisms, in the way that embryonic development has become an unconscious series of solutions to physiological and other problems which were once to a large extent new problems at the end of development.
It would seem to follow, although Schrodinger makes no reference to Waddington’s concept as such, that genetic assimilation may be linked with conscious processes; that is to say, whatever the mechanisms of genetic assimilation may be, the process is in part correlated with, or follows upon, phenomena of consciousness. According to the parallel drawn earlier, it would seem to follow also that the mechanisms of new-gene acquisition may be related in a similar way.
This perspective is not necessarily out of keeping. with the principles of synthetic evolution theory and the “DNA dogma.” Under these complementary viewpoints, the rise of the capacity for conscious experience is supposed to have occurred through natural selection, insofar as increases 1n this capacity have been of adaptive advantage. Although evidence may be lacking, it is also implicit in these viewpoints that, as the capacity for conscious experience has increased during evolution, there has been a corresponding increase in the quantity of genetic material and information on which this capacity is dependent. The anagenetic viewpoint adds to this merely an emphasis on the role of genetic
information, or, in terms of the analogy used earlier, on the importance of new cards in the deck, over and above the
importance of the reshuffling and mutation of old cards. A completely deterministic theory consistent with all these viewpoints emerges, for example, from the hypothesis that there is in the gene pool of every species undergoing anagenesis, a relatively small complement of new and untested genetic information, perhaps derived largely from imperfect or mutated tandem replications of older genes, which is selected and becomes established in the pool to the extent that it determines adaptive responses under recurrent conditions in the course of generations. Under such a hypothesis, all conscious responses, as well as physiological and behavioral responses, are completely predictable in principle to the extent that the quantum mechanics of DNA may be predictable.
The validation of such a deterministic theory would resolve the questions of the mechanisms of new gene acquisition and of genetic assimilation, and of the relation of these processes to the phenomena of consciousness. The experimental difficulties with the pursuit of this validation are obviously enormous, but beyond these, there appear to be insurmountable theoretical difficulties. Thus, even assuming resolution of experimental difficulties to the point that biopsies could be made which would quickly reveal DNA structure to the last nucleotide, there would inhere an element of unpredictability in its structure, hence in its function, due to the limitations on observation posed by the Heisenberg uncertainty principle. Furthermore, in respect to what the DNA may be determining at a given time, Bohr (1933) has pointed out in a biologica1 extension of his complementarity principle that, as one attempts to pursue observation of life processes to ever deeper submicroscopjc levels, he must interfere increasingly with the life of the organism, until in the limit he must kill it to observe at the molecular level, if not at grosser levels. .
This argument perhaps applies with even more force to the investigation of phenomena of consciousness than to the investigation of behavioral and pysiological phenomena. Additional theoretical difficulties which apply to ‘ the problem of validation of deterministic biological theories have been pointed out by Elsasser (see 1961).
These difficulties mean that there is no possibility of ultimate scientific elimination of all hypotheses alternative to those of synthetic evolution theory and the DNA dogma. Thus, if we were to know the DNA, down to its atoms, still we could not know every potentiality of the organism, even though every environmental eventuality were taken into account. The operation of factors other than genetic information in the determination of biological phenomena therefore cannot be written off. Other factors might be either deterministic or indeterministic: for example, as an alternative deterministic factor, it may be postulated that conscious response, if not other processes, is partially determined by the matter-energy of food, light, etc., entering into metabolism during the response process in an active rather than passive way.
As an example of an indeterministic factor, one may postulate that the indeterminacy of the Heisenberg principle is inherent in nature as well as being an operational limitation on the possibilities of investigation. The consideration of such alternative possibilities is particularly relevant to the questions of new genes and new
terminal phenomena, which involve the greatest uncertainties and greatest difficulties of validation for the synthetic and DNA theories–hence this discussion. If alternative factors operate at the level of conscious response, they may also operate at lower levels of behavioral and physiological processes, etc.; at least in the case of new behavior patterns or physiological effects where, according to the above suggestions, new genetic information and the process of genetic assimilation are likely to be involved.
Thus the phenomena of genetic assimilation may involve the operation of such alternative factors. Supposing that they do, the possibility exists that the alternative factors play a decreasing role, while genetic determination becomes increasingly dominant, in
the series of phenomena from one’s present conscious responses and perhaps other more or less novel processes at one end of development, toward the increasingly rigidly determined phenomena at the other, embryonic end.
The operation of alternative factors would tend, insofar as it applies, to relegate the genetic material to a somewhat passive role, analogous to that of a memory chip in a computer, for the storage of coded information dealing with problems already solved. The impossibility of eliminating these alternatives means then that, in effect, one cannot say whether life is DNA’s way of making more DNA, or that DNA is life’s way of making more life, where the term “life” has special reference to phenomena of consciousness. (I am once again indebted to Professor Danforth for a succinct statemen~ of the issue.)
Perhaps the element of caprice and unpredictability which is experienced in some phenomena of conscious response in the here-and-now, can be drawn up in favor of the operation of the alternative factors along with genetic determination. The complete determinist position is of course logically impeccable, but the fact is that we do not experience complete determinism. Prom the empirical stand- point, therefore, the indeterminist position is defensible as being in better accord with reality. It follows that there are inherent limitations on the possible accomplishments of science; this and related points are discussed further in Part II.
On the other side of the question, anagenetic evolution of plants may be drawn up in favor of the DNA dogma, with perhaps some reservations. More fundamentally, however, the admission of alternative factors involves a sacrifice of theoretical unity, with no hint in sight, for example, as to how determination by matter-energy in metabolic transit could be transferred to genetic determination through a process of genetic assimilation. It is primarily for this reason that an adherence to the principles of the synthetic and DNA theories is implicit in this work as a whole.
12. LONG TERM IMPLICATIONS OF ANAGENESIS.
The most reasonable extrapolation into the distant future would appear to be one which assumes continued anagenesis, resulting in ever more intricately organized organisms, societies, ecosystems, etc. Aside from the general implication that man is not here to stay as such, and stands to be replaced by forms which may regard him as he now regards the ancestral tree-shrew–a point not without moral and philosophical overtones–the nature of this higher
organization is to a large extent unpredictable in principle, much as the human condition in this technological era was unimaginable even a few centftries ago.
It is not altogether too far-fetched, however, to consider such possibilities as homeostasis between biotae of planets with intelligent life distributed throughout the galaxies, as natural results to be expected of continued anagenesis. These considerations will become advisable, in fact, if and when attempts now in progress to exchange signals with life on other worlds succeed. It is not pertinent to explore these matters here, but it is pertinent to examine, at least briefly, the taxonomic consequences of long-continued growth of the tree of life.
a. “Dynamic taxonomy.”
A dilemma arises from the evolution of ever-new taxa from existing gene-exchanging units as the tree of life grows. The present discussion is restricted to the evolution of multicellular plants and animals. Speaking from the standpoint of diversity alone, i.e., neglecting organizational divergence, today’s species tend to become tomorrow’s genera, today’s genera tomorrow’s families, etc. From this standpoint, therefore, ranks of taxa should be raised as time passes and diversity of the taxa increases, so that, for instance, the mammals should become a phylum in time, the chordates a kingdom, the metazoans perhaps an empire, etc.The invention of new names of categories at the high end of the scale is eventually necessary under this view.
From the standpoint of divergence, however (neglecting diversity), such a procedure is objectionable. To a degree divergence increases with time, to be sure, but the divergence, for example, of mammals, birds, and reptiles from one another, nonetheless cannot achieve the degree implicitly accepted as necessary for phylum distinction. In no small measure this is to be explained by the higher level of organization of the common ancestor of the amniotes, as compared with that of the ancestors of the various phyla.
If we regard the known or postulated level of organization of common ancestor as the guiding criterion for the ranking of plant and animal taxa, the complications associated with divergence are avoided and we arrive at a conclusion diametrically opposite from that indicated by consideration of diversity alone; ranks of taxa should remain fixed, and new categorical names need to be invented at the low end of the scale as time progresses.
This dilemma is not pressing, mainly for two reasons. First, it requires time of the order of a hundred million years before any effect on the high end of the scale can be fe1t. Meanwhile, the .elevation of species to genera and genera to families, even families to orders, might be carried out as radiation demands, without serious crowding of the existing scheme at the high end of the scale. It is largely
the quasi-geometric increase of ages of categories which makes this so (see Figure 2).
Second, the “noise” caused by differences of evolutionary rate in diverging lines, among other factors, has so great an effect on the designation of taxonomic rank that the general trend of increasing divergence and diversity of taxa, i.e ., the orderly “signa1” tends to be obscured. A classic case in point is that of crocodile, lizard, and bird (Bigelow 1956), where we find a separate class (the birds) more closely related in time to the crocodiles than are the lizards, although crocodiles and lizards remain grouped.in the same class on account of their greater retention of similarities.
While thus not pressing, the dilemma is real and is of theoretical interest. The Linnaean scheme may be described as a “static” taxonomy, in the sense that its hierarchy of ranks serves for the classification of organisms .as they exist at this moment in evolutionary time, but was not intended to serve for classification at times in the long past or future . (No reflection on the science of bio- systematics is intended in the use of the word “static,” this science on the contrary being quite fast-moving today.) A ttdynamic” taxonomy may be defined as a system especially designed to be applicable at all times, or regardless of time, for the classification of the world’s organisms. The admission of ever-new categorical levels as the tree of life grows is a demand which falls upon any dynamic taxonomy. As indicated above, the new levels might be introduced at the high end or at the low end of the hierarchy. The introduction of ever-new ranks at intermediate levels is also possible, and indeed, the introduction of new levels into the static Linnaean system necessitated as groups have become better known, has followed largely this pattern.
It can be argued that there is no need for any other practice in the development of a dynamic system. However, this practice seems objectionable from ·an idealistic standpoint. Perhaps the idealistic objection is essentially that such a practice does not lend itself to mathematical description. Another possibly stronger objection emerges from the effect of level of organization on the relative divergence of lowest-ranking taxa (see below).
Let it be assumed, then, that the introduction of new levels is to be at either the high or low end of the scale. To add at the high end is by implication to employ diversification as the main guiding principle in ranking, which is objectionable on the basis that it is in fact used as a subsidiary· principle (section 4). To add at the low end is to use level of organization of common ancestor as the main guiding principle. This is objectionable where noise is louder than signal, so to say, but offers a fixed base of reference for ranking, albeit not easily determinable. This fixed base of reference (the level of ancestral organization) is moreover an index of taxonomic potential, in that the higher the level, the lower the relative divergence of the descendant taxon, therefore the lower its relative rank.
In a word, no matter how old the child may become, he can never become as old as his father, nor can he attain to as simple an origin, except perhaps through neoteny.
The use of level of organization of common ancestor as the principle criterion of taxonomic rank has the consequence, of course, that least diverse taxa in modern times, for example, would be assigned a lower rank than least diverse taxa in the Proterozoic. This change of procedure would have a measure of justification on the following grounds. Let it be assumed for the sake of argument that speciation requires the accumulation of a given mean number of non-homologous genes in the diverging populations, and that this number does not change greatly with advancing level of organization from one era to another. Then it would follow that least diverse taxa show decreasing relative divergence as time goes on–that adjacent species were relatively more divergent in the remote past and will become relatively less divergent in the remote future–because the proportion of genes-in-common would increase with the rise in level of organization of common ancestors.
It is interesting to consider the bearing of this argument on the concept of species. On the one hand, adjacent species may be recognized as units distinguished from one another on the basis that they look different to a certain extent. The extent is not defined, but it cannot be too low or the units will be recognized as subspecies, say; likewise it cannot be too high, or they will be recognized as genera. This implies that the species concept includes relative divergence among its criteria, and in this respect the practice of assigning new categorical ranks to new 1east-diverse taxa as they arise in time, as suggested above, would be defensible.
On the other hand, the species concept involves ecological and genetic factors which may have been more or less unaltered throughout the greater part of evolution, although this is open to question. From this standpoint, therefore, it is perhaps advisable to retain the terminology of species and infra-specific categories, and in respect to adding categories at the low end in accordance with the idealistic approach suggested here, to.do this at a level
just above species. The potentially endless series of new ranks necessary in the long run suggests the advisability of designating rank by number rather than by name. Names already in use need not be abandoned, but could be coupled with a Tx notation, where x is level of organization of ancestor. This would be similar to Beckner’s Tj. notation based on set theory; with two notable differences: a) x decreases while j increases with taxonomic rank (this to allow for indefinite increase of the subscript with time), and b) x need not be integral. A numbering system would also be necessary to distinguish taxa at a given level (given value of x) if the system is to be independent of names, and this might be done with pre-scripts or superscripts. Such questions arise with the pursuit of a dynamic taxonomy, but cannot be explored here. Not least of them, however, is that of the quantification of the concept of level of organization, on which Part II has a bearing.
PART II. INQUIRY INTO THE PHYSICAL MEANING OF ANAGENESIS
13. DEFINITIONS.
The term “anagenesis,” in the sense in which it is used in this work, was introduced by Rensch in 1947. Its literal English translation is “development in an upward direction” (Rensch 1959:97). “Anagenesis” was distinguished by Rensch from “cladogenesis” (“branching development”), the latter being essentially equivalent to “adaptive radiation.” Huxley (1957) endorsed these terms, proposing a generalization of ”’anagenesis” to include all types of biological improvement, from detailed adaptation to general organizational advance; Rensch had excluded special adaptations which do not provide a lasting advantage.
Huxley suggested also a third term, “stasigenesis,” to describe the kind of evolutionary stagnation observed in Lingula and many other so-called “living fossils.” Hence there was proposed a word of similar etymology for each of the three major evolutionary processes: cladogenesis, leading to “clades” (taxa; see, however, section 1); anagenesis, leading to “grades” (integrative levels or levels of organization); and stasigenesis, leading to living fossils or relicts. The introduction of this new terminology has not been greeted without protest (cf. Straus 1958), but Simpson (1961) admits the advisability of some convenient terminology, although he prefers a different one.
Simpson’s “progression” is equivalent to “anagenesis” sensu Huxley, except that it includes also degenerative changes; his “splitting” is essentially the same as “.cladogenesis;” and his “equilibrium” the same as “stasigenesis.” Simpson moreover considers that there is a fourth major feature of phylogenetic patterns, and he introduces another word for it: “termination,” essentially equivalent to “extinction.”
Simpson’s “progression” is a more general concept than “anagenesis,” but precisely for that reason it is less suitable for. the description of that salient feature of evolution which is our subject of inquiry, the increase of complexity and organization from naked genes to modern high forms, wh1ch is not represented by degenerative changes. There is, furthermore, an element of contradiction in including “regressive” changes under “progression.” For these reasons and others, including sound etymology, Rensch’s term as generalized by Huxley has been adopted here.
Circumstances have not necessitated the use of “cladogenesis” and “stasigenesis” here, but it may be remarked that, aside from sound etymology, these terms have in their favor a measure of symmetry, as it were, and in the balance this may outweigh the disadvantages of length and unfamiliarity.
Rensch (1959:281-301) has considered the historical development of the concept of anagenesis, some of the cardinal anagenetic events in the history of life, and the major ways in which anagenesis is manifested. The main purpose of Part II is to try to carry the analysis of the concept to a deeper and more general level, where it can be understood to a degree from physical principles. Rensch’s analysis is summarized first, with a few comments. I have
tried in the prefatory remarks to the further analysis, to explain why the’ problem is of central interest and importance for biological theory.
14.”CLASSICAL”MANIFESTATIONS OF ANAGENESIS.
From his historical ·review of literature on progressive evolution, Rensch gleans the following major characteristics of anagenesis:
1) Increase of complexity. This refers especially to differentiation of cells, tissues, and organ systems. It deserves mention that such differentiation is ‘manifested in the ontogeny of the indi-vidual organism, as well as in its history. Extrapolating by analogy, increased’complexity of societies and ecosystems might be included here, though Rensch does not do so.
This increase of complexity is perhaps the most widespread and clearly evident characteristic of anagenesis, and is accordingly the one which has been longest recognized. It is implicit in the Scala Naturae much in vogue in the 17th and 18th centuries and probably traceable to Aristotle’s thought, which is to say that the factor was recognized long before the evolutionary interpretation of the sequences of living forms (cf. Eise1ey 1958). In modern terms, increased differentiation is presumed to have been achieved as the result of different selective forces acting on cells differently situated. Rensch notes that it is especially abetted by increase of body size in accordance with Cope’s rule, which provides more cells, therefore more situations for differential action of selection. He notes further that mutations causing allometric growth can in principle explain much of the differentiation resulting from response tu such selection, but that secondary simplifications and chemical differentiations are also among such responses.
2) Increased rationalization [economization]. Included here is division of labor among cells, tissues, etc.; secondary simplification with increased efficiency and improved engineering, as in the reduction of digits in ungulates; “interiorization” and centralization, as in the evolution of the vertebrate central nervous system. This concept is apparently a distillation and synthesis from factors emphasized by several authors, both pre- and post- evolutionary (Goethe, Bronn, Darwin, Haeckel, Gregory, and others). In respect to simplification this factor might appear to be partly in conflict with the first, and even with itself; but improvements in engineering accompanying secondary reduction of parts may typically result in a net increase of complexity, at least at the genetic level, in
which case there is no conflict.
Note also the interconnection between increase of complexity (item 1) and division of labor (item 2), making the separation of the two factors somewhat artificial.
3) Special complexity and rationalization of nervous systems.
This is but a repetition of the first two items, applied to nervous systems. As such, it is stressed by Rensch because of the evolutionary prevalence and importance of what he aptly calls “brain victories.”
4) Increased plasticity of structures and functions . This is a concept developed by Rensch himself, to a greater degree than are the others. The point is that many organs and organ systems (e.g., salivary glands, pancreas, liver, homeothermic mechanisms) have become functionally more versatile in the course of evolution. The increased freedom of mobility of neck, tongue, and limbs (notably Primate forelimbs) in higher vertebrates provides another interesting
type of example.
To a degree, there is a contradiction of the trend toward increased specialization and division of ,labor here, but as Rensch recognizes, there is apparently always an underlying increase of homeostatic mechanisms, and it is this which perhaps lies at the heart of the anagenetic nature of the characteristic in question. In application to the sensory and nervous systems, Rensch points to the increase of regulatory mechanisms in the evolution of the vertebrate eye, and especially to the intricate homeostases implied in reflexes, still more in learning, and most of all in insight (known in psychology as “aha” behavior). Although Rensch makes no specific extension of the notion of increased plasticity to higher integrative levels such as society and ecosystem, the underlying feature of homeostasis suggests this extension, and the examples of comfort-seeking and migratory behavior which he provides point in that direction.
5) Improvement permitting further improvement. This factor has been stressed earlier by Simpson as “progress in adaptability, “ and by J. S. Huxley as “non-restrictive improvement.” It may be regarded as a special type of increase of plasticity, or perhaps better, as a general increase of plasticity, but is of such importance in Rensch’s view as to deserve separate listing. It represents the opposite of blind-alley specialization, and as such is a necessary feature of anagenetic evolution by Rensch’s definition, whereas Huxley’s allows for the
inclusion of blind-alley as well as non-restrictive adaptations under anagenesis. Note that the view of this factor as a special case of the previous one suggests again an increase of homeostatic mechanisms as the underlying principle.
6) Increased independence of the environment and increased autonomy. This factor was perhaps first emphasized by Herbert Spencer, and subsequently by many authors, including J. S. Huxley and Simpson. It is often invoked in discussions of the evolution of the human condition, and is generally familiar for that reason. Emerson’s statement quoted above, that the fluctuating external environment of one level of organization tends to become the homeostatic “milieu interieur” of the next, is relevant in the consideration of its meaning, and points to a somewhat broader interpretation than the usual. There is again a rather ‘obvious overlap with previously listed factors, and an implication of increased homeostatic mechanisms as the underlying principle.
7) Regression. Rensch’s inclusion of regression is inconsistent with either his or Huxley’s definition of anagenesis in my interpretation, and is more in line with Simpson’s concept of progressive evolution. Rensch distinguishes non-restricted regression, illustrated by the evolution of internal parasites, from restricted regression, illustrated by limb-reduction in lizards, etc. There can be no question, in view of the prevalence of vestigial characters, that restricted regression is often advantageous. This can be explained quite generally on the principle that any unused character constitutes a metabolic burden, and the selective advantage will accordingly lie with variants wherein this burden is reduced; it is of interest that Darwin conceived this principle but did not himself apply it consistently. It is true that mutation pressure alone could produce regression when selection pressure is relaxed, but the factor of metabolic burden is always present to provide definitely negative rather than merely zero selection pressure .
The concept of non-restricted regression is somewhat more problematical: the general regression of adult structure in parasites is typically accompanied by increased complexity of the life cycle as a whole, so that it becomes difficult to say whether there has been a net decrease in organization at the genetic level. In any case, regressions are not given further consideration here, on the ground that they do not truly represent anagenesis.
It will be observed that the.items chosen for listing by Rensch are somewhat arbitrarily separated from one another. Taken together, however, they provide as good a summary of biological thought on the ques:ion of anagenetic evolution as any known to me. Even so, it should be noted further that the list cannot claim to be complete. One item omitted is that of the evolutionary tendency toward enlarge- ment of the organism’s world, expressed by Beurlen (1937; .see Rensch 1959:284) as an “Umwelterweiterung;” related to this and perhaps more fundamental is what may be called a “Merkwelt-erweiterung,” stressed recently in terms of an increase in “richness of experience” by Waddington (1960b)– some discussion of this .point is included below. Other items familiar to evolutionary biologists” such as tagmosis and anisomerism, can have a place in Rensch’s list.
It does not follow, however, that there are not perhaps other important omissions.
15 . THE BEARING OF ANAGENESIS ON BIOLOGICAL THEORY.
The theorist’s desire for a comprehensive understanding of life which meshes with our understanding of the physical universe is the prime motive for the present pursuit of the fundamental basis of anagenesis. The pursuit has led to a consideration of information theory, which has been linked with physics (cf. Brillouin 1956). I have tried to delineate the limitations as well as the comrehensiveness of this approach, exploring in a tentative way its bearing on phylogeny, ecology, and some topics outside biology proper, wherein the human condition comes under discussion.
The aim in attempting to embrace biology within physics is essentially the same as that in seeking a common theory of light and electricity, and of·heat and bodies-in-motion. The successes of physics in including ever-larger and more diverse features of the environment within ever fewer and deeper, more comprehensive theories has inspired the quest for a single viewpoint from which all manifestations of nature can be understood. Life ranks with light, heat, electricity, and matter as one of the prominent classes of phenomena in nature. The ultimate aim of theoretical science is, accordingly, a unified theory which embraces all biological as well as physico-chemical phenomena.
One point, essentially true of all theory, deserves special emphasis in respect to biology. A perfect theory would be one in such complete accord with nature that accurate predictions could be made from it, to any future time; relativistic celestial mechanics approaches such perfection. It is recognized now that no absolutely perfect theory is possible even in principle, developments
in physics in the 20th century being largely responsible for this recognition. The working hypothesis of science has been accordingly somewhat modified: perfect comprehension is taken to be indefinitely approachable, but no longer is it thought to be actually attainable, even in principle. In regard to biological theory, this comes home in a way which points up the significance of the problem of anagenesis for science in general.
If a perfect theory of life were attainable, a scientist in possession of it would be able to predict all behavior and thought of living things, including his friends and even himself. The facts of personal experience of life are that such complete understanding does not exist. An investigator may proceed on the assumption that the impossibility is not a matter of principle. But one may equally well proceed, with the physicists, on the assumption that perfect understanding is only asymptotically approachable, that, in effect, nature always reserves some “room for play.” The desolation of personal existence which the attainment of perfect understanding as described would entail is, however, no reason to take the physicist’s course. A real reason to do so is given by the empirical fact that our degree of understanding decreases as we turn from strictly physical phenomena to entities at levels of organization more nearly equal to our own.
This is not to say that’ the degree falls to zero when one turns to himself, or even when he turns to the phenomena of culture; it is merely to say that it falls. It may be added that it is not to be expected on logical grounds that a system at a given level of organization could process sufficient data for the complete prediction of another system at the same or at a higher level.
The grounds for a physical theory of life have been laid largely in quite recent times. The accumulated knowledge is now so enormous that it is difficult to delineate even the foundations without doing injustices. Few will dispute, however, the key roles of Darwin’s theory of evolution on the one hand, and the elucidation of the biochemistry of the cell on the other. Genetics has linked these insights, so that we can now view biology as the evolutionary chemistry of certain macromolecules, much as organic chemistry came to be viewed as a branch of the chemistry of carbon a century ago. To be sure, this view of biology is an oversimplification, but all scientific generalizations are in some respects oversimplifications,
even the most exact.
The question is, rather, whether this view is the right one in its essentials. Skeptics there are, as noted above; yet, it carries enough conviction to be widely accepted as a working hypothesis. It accords with previously drawn generalizations of science as a whole, notably the atomic theory and its multiple consequences,including all of chemistry; and it carries further the undermining of anthropocentrism which has characterized science since Copernicus. It underlies the investigations into the “coding problem” (relation between nucleic acid and protein “alphabets”), mechanisms of protein synthesis,
nature of enzyme activity, and others among the most fundamental investigations currently under way.
From this viewpoint, evolution is the result of natural selection acting on effects of random changes in the coding of replicating nucleic acid molecules (genes). From the outset of selection theory, Darwin and Wallace stressed that natural selection entails what we ‘are calling anagenesis, in that variant offspring better organized in any way will have, as a rule, an advantage over competitors at the old level of organization. The biochemical theory of the gene makes possible a molecular interpretation of anagenesis: variant genomes with more encoded information of adaptive advantage will, as a rule, increase in relative numbers in the genome pool of the biota.
Some difficulties with this interpretation were discussed in section 11, and an alternative hypothesis formulated, to the effect that only established methods and solutions to problems are coded on the genetic material, the capricious element in response deriving from input matter-energy. There is apparently no known procedure for testing these alternatives; indeed, viewing them from the standpoint of the nature-nurture controversy, a complete resolution is not to be expected. The conflict may center on the question, can DNA be the basis for autonomous behavior?
It would seem that autonomy is inconsistent with the postulate of a fixed nucleotide sequence as determinant of all characters of the organism. As pointed out earlier, to deny autonomy is the
theoretical means of escape for the determinist; but an element of caprice is the fact which we experience, i.e., there is unpredictability in behavior. Be that as it may, the hypothesis that biology is the
evolutionary chemistry of macromolecules, notably DNA, is adopted in what follows. Although it is possible that to take this stand is to study the bath to the neglect of the baby, it cannot be denied that there is much to study in the bath.
16. A PHYSICAL INTERPRETATION OF ANAGENESIS.
The coded genome has been likened to an “aperiodic crystal,” and life to a growth of such crystals (Schrodinger 1944). This idea accords with the necessity, because of its stability throughout many replications, that the hereditary material is matter in the molecular state, in which the Heitler-London forces derivable from quantum mechanical principles hold the atoms and molecules together against the thermodynamic forces of random bombardment. Schrodinger distinguishes two types of physical law, governing two classes of phenomena or, it is useful to imagine, two extreme conditions of the universe: 1) mechanical law (more fundamentally quantum mechanical, since macromechanica1 law is but a limiting case), and 2) thermodynamical law, governing such phenomena as diffusion and heat flow, which are irreversible.
A universe at absolute 0 0 K would correspond to the extreme
condition in which only mechanical law prevails; frictionless clockworks represent this state on a small scale, the solar system approximates it on a larger scale, with respect to mvements of the planets and satellites. A universe of· gas so hot that even its atoms were stripped down to bare nuclei would approach the other extreme, in which thermodynamical law prevails; the interior of stars is believed to approximate this condition, and “plasma” represents an approach t it on earth.
Life of course exists in an in-between condition of nature. Schrodinger sought to emphasize that life must be based on mechanical laws, i.e.., he wished to see it as a branch of quantum mechanics; hence, the analogy with crystals. And indeed there is fundamental insight into the place of biology in the physical “Weltbild” in the view of life as an aperiodic crystal growth, knowing the intended quantum mechanical implications of “crysta1.”
This view lends a physical interpretation to the biochemical-genetic view that life is “DNA’s way of making more DNA.”
These insights may be necessary for a theory of life, but as is to be expected they are not sufficient. Perhaps their greatest shortcoming is in leaving out of consideration the matter- and energy-turnover essential to life. Inorganic crystals do not grow by means of such a turnover, but as a rule, by simple cooling, i..e., as a result of the environment’s approaching that absolute zero condition of pure mechanical law.
The aperiodic crystals of life, however, grow only in an environment of essentially constant temperature (from the standpoint of the spectrum of temperatures as a whole, the life-range is nearly constant), and then only if they have access to sufficient supplies of energy for metabolism. This process of metabolism involves laws of the thermodynamic type, such as -diffusion, in fundamental ways. Both kinds of laws are therefore of concern in what follows.
Consider the macroscopic aspects of life, viewed as an aperiodic crystal growth. Most outstanding is the fact that life is manifested to a great extent in large and very complicated organismic forms, far removed from naked genes in a dilute organic soup. It is a general observation that all such organisms arise from very similar parent organisms. There are some seeming anomalies, such as the development of caterpillars from the eggs of butterflies, or of fern gametophytes from spores of the sporophyte. But when the whole life-cycle is taken as the reference form, the principle holds very generally, that any given’ biological form arises from a closely similar pre-existing form.
Weizsacker (1949) has pointed out that this principle is equally applicable to inorganic forms, notably the stellar and nebular forms of astronomy, which have been his special concern. In virtue of its generality, he has seen fit to give it a name, the “principle of continuity in the origin of forms.” At the microcosmic level–where occur such phenomena as the “annihilation” of elementary particles with the disintegration of atomic nuclei, i.e., the radical and complete. transformation of matter into radiation, and vice versa, with no evidence of gradual transition–Weizsacker’s principle may find exception. But such transformations have to do with entities of relatively low complexity compared to that of forms observed directly, on which the principle is based; they do not detract from the validity of the principle in the range of forms at levels of organization with which we are concerned in evolution.
Anagenesis in biology, as the process by means of which there are derived increasingly complex and highly organized forms from simpler ancestral form3, may thus be regarded as corollary to Weizsacker’s general principle. The physical interpretation of anagenesis turns on the connection of this qualitative principle with quantitative physical principles. This connection has not been developed very far, but Weizsacker has clearly indicated its nature. The principle is viewed from the standpoint of statistical mechanics, from which it takes the aspect that highly improbable states of matter’-energy can arise only from closely similar states.
It is of course Boltzmann’s interpretation of the second law of thermodynamics which suggests this approach: the degradation from usable mechanical or electrical energy to unusable heat energy (the increase of entropy) in a closed system is the result of the system’s inevitable assumption of increasingly disorderly or probable states as the constituent atoms and molecules become disarranged, much as the cards in a new deck become disarranged with shuffling.
Just as there is continuity between successive states of physical systems in the course of this degradation (as in the wearing out of machinery or the lysis of dead cells), so the anagenetic appearance of higher forms involves a continuity between successive states.
The evolution of life toward increasingly complex forms was early seen to be in apparent opposition to the general trend of physical systems toward a “heat death” in accord with the second law. The Boltzmann interpretation of the second law, together with the similar interpretation of Weizsacker’s principle, makes possible a better statement of the apparent conflict: evolution has resulted in the appearance of increasingly improbable states of matter-energy, while closed physical systems, and presumably the universe as a whole, have tended toward ever more probable states.
The history of the conflict involves some erroneous physical thinking (see, for example, the review of theories based on a “principle of stability” as a cause of evolution, by Holmes 1948). The current consensus, if any, is perhaps best represented by Blum (1951), who reexamined the question and came to an entirely negative conclusion regarding violation of the second law. Blum” s resolution of the paradox rests on the argument that there is more entropy-produced during metabolism by living material than negative entropy captured and stored; i.e., metabolism involves, irt the balance, more molecular disarrangement than arrangement. Hence the second law presumably holds in any closed system capable of supporting life.
Generalizing Blum’s argument to the whole evolutionary scene, this implies that the amount of entropy produced by the degradation of chemical and sunlight energy by the biota since its origin, is greater than the sum total of all order which the genetic material has succeeded in collecting throughout evolution, manifested in structural differentiation and homeostatic mechanisms as outlined above. Verification of such a generalization is obviously a difficult matter, however.
The apparent conflict of anagenesis with the second law can also be expressed in terms of information theory. This approach is of interest, since many authors have been influenced to use information theory in the corisideration of a variety of biological problems (cf. ‘Bril1ouin 1949 et seq.; Quast1er 1953; Rothstein 1958; Yockey 1958; Patten 1959; Schma1hausen 1960). The approach rests on the fact that the mathematical definition of information, originally developed for the expression of the information content of messages as a concept useful in communications engineering, has the same mathematical form as Bo1tzmann’ s statistical mechanical definition of entropy; thus, information in “bits” is proportiona1 to the negative of entropy (“negentrapy;” Brillouin 1951) in thermodynamical units, e.g., ergs per degree centigrade. The constant of proportionality being very large (ca.1016), a numerically great gain in bits of information corresponds to a small decrease of entropy in ergs/0C. Brillouin (1956:289) has estimated the structural negentropy of the entire network of the American telephone system, and found it, negligible compared to the entropy of the constituent parts, from which it follows that if the whole network were torn asunder and the parts piled up as junk, the net increase of entropy would be relatively slight. As Brillouin indicates, this is probably not true of the men who built the network, although we do not yet know how to
calculate the negentropy of living organisms satisfactorily.
This result has the following intuitive interpretation. To a first approximation, the information content of a message in bits corresponds to the number of symbols in it. In ,a physical system, the number of symbols in a message’ specifying its exact state is of the order of magnitude of the number of molecules within it. The complete specification of the state of any macroscopic system therefore requires an enormously long message. The message necessary for the construction of the entire American telephone system constitutes but a minute fraction of the message necessary for the complete molecular specification of the system; hence, there is proportionately negligible negentropy or information difference between the functioning system and the pile of junk.
It may be mentioned 1n passing that as technology
advances, for example in the areas of solid state physics and microminiaturization, the proportion of the available negentropy of materials actually utilized in the function of devices made from them tends to increase. There is hardly any question but that much progress in this direction lies in the future. Whether conscious attention to the underlying negentropy principles involved would make a difference to this progress is an open question. This is possible, however, and to that end, what is outlined in section 17 below may be suggestive, some indications being given there
as to the manner of increasingly efficient use of the negentropy of. materials and natural processes by genetic material during the course of its evolution.
The second law in terms of information theory states that the information which is attainable from or about a closed system never increases, and tends to decrease in the course of natural processes. A highly ordered condition of the system entails that the range of states in which its atoms may reside is small compared to the total range of states open to an unrestricted equivalent set of atoms in nature. The information which in principle can be attained about a system is for some purposes better expressed as what can be determined about the states which it cannot be in, than as what can be determined about its actual state. Thus the attainable information corresponds to the range of states which can be excluded, and this range tends to decrease in the course of natural processes, as atomic order gets disarranged.
Perhaps Rothstein’s (1958) statement of the second law in terms of information is most fundamental and succinct: no information can be extracted from pure noise.
A more detailed discussion is to be found in Brillouin (1956:152ff.). In informational terms, the seeming contradiction of the second law is that anagenesis entails the evolutionary appearance of systems containing ever-increasing quantities of attainable information. Its resolution takes the form that over-all information loss through the degradation of energy during metabolism is presumably greater than local information gain by collection and storage in the living organism; the second law therefore prevails.
This explanation can obviously be extended in principle like the other, to the domain of the biota in the earth-sun-space system since the origin of life. Verification at this level of generalization is, however, no easier in informational than in other terms.
Anagenesis, then, is interpreted from the standpoint of information theory as the evolutionary accumulation of information or negentropy in biological systems. Insofar as sound, this interpretation has the advantage that it render anagenesis a physical concept. There is, moreover, no obvious difficulty with the incorporation into this concept of older biological ideas of anagenesis (section 14); structural differentiation and homeostatic mechanisms involve information, whether from the standpoint of instructions required for their formation, or from the standpoint of information attainable from or about the systems possessing them.
Anagenesis may also be spoken of in terms of rising levels of organization, which fact raises the question as to how to define “organization” in terms of information. Rothstein (1958:34ff.) has suggested a definition, such that the degree of organization of a set of elements is measured by the difference between the sum of their entropies assuming no interactions, and the sum when interactions are taken into account. This difference is the amount of information introduced into the set because of the interactions; hence organization is indeed information. This definition is perhaps too simple (cf. Muses, in Rothstein 1958:lxxviii), and its application to biological systems, particularly to their evolution, appears to be remote. Yet it suggests a sound approach, and might be coupled with Weizsacker’s (1949) point, that the number and variety of forms within a form reflects its level of organization, to provide at least a
tentative answer to the question.
Extending the informational interpretation to the level of molecular biology, anagenesis implies the accumulation of information or negentropy in the sequence of nuc1eotides encoded on the DNA molecules of the genomes. This extension carries the problem of anagenesis into the arena of the coding principle, which has presented other problems, intensively investigated because they hold forth the promise of a synthesis of molecular-biological with physical principles.
It might be argued that there is an obstacle to this synthesis in the fact that the mathematical definition of information attributes as much information to a nonsense message as, say, to a declaration of war of equal length. This is not a real obstacle, however. Information theory takes it for granted that sender and receiver are attempting to communicate in a common language, and that messages are sent in meaningful form with attempt to avoid nonsense. Under these terms the extraction of the operational meaning of the message by the receiver is also taken for granted.
A second difficulty may be more serious. The role of information as basis for action and decision is not part of the mathematico-physical theory of information (cf. Brillouin 1956). Since decision and action are at least partly unpredictable, in one’s self as well as in other beings at a comparable level of organization, it may be that this aspect of information is partly unresolvable in principle. Bohr (1933) and Elsasser (1958,1961) have pointed to other and perhaps overlapping difficulties with the physical interpretation of biology as noted earlier. A need for caution is indicated by these obstacles, but they do not yet, at least, call for abandon of the approach through information theory.
For the most part, discussions of the relation.between biology and physics have not dealt specifically with the problem of anagenesis. Weizsacker (1949) has illustrated how a seemingly more highly differentiated form can arise from a seemingly less highly differentiated one, even without local capture of information: his example is that of the circle of balls derived from a pile of balls rolled onto a plane from the top of a radially symmetrical hill. He explains that the energy content of the’balls, before and after, must be taken into account in appraising the change in entropy content of the respective forms. The point is not to be denied, but it is difficult to see how it can fully account for the evolution of modern genomes from naked genes, for example, where a capture of information or negentropy is strongly implicated.
Rothstein (1958:82ff.) presents ,an interesting argument for the inevitability of anagenetic evolution, evidently based on a theorem of Prigogine from the thermodynamics of irreversible processes (cf. De Groot 1952, where the theorem is generalized and described as an extension of Le Chatelier’s principle). According to this theorem, the rate of entropy production tends toward a minimum in open or non-equilibrium systems. Rothstein interprets this to mean that in a system consisting of a star as high-temperature reservoir and interstellar space as low temperature reservoir, an intermediary body such as a planet, which may be viewed as a heat engine operating between these reservoirs, must evolve toward minimum conversion of input energy directly into heat, and maximum conversion into high-grade energy. Rothstein infers that life or other highly organized matter is “far more ubiquitous in the cosmos than heretofore thought possible, for the above considerations would apply to cases where life as we know it is impossible” (p.87).
Although I am unable to evaluate this argument. conclusively, it appears to me that it is going beyond the limits of strict applicability of Prigogine’s or the extended Le Chatelier’s principle to invoke it in an argument involving biological evolution. The essential requirement for such a phenomenon, I submit, is that quantum mechanical and/or other laws of intricate organization of the microcosm prevail. This is Schrodinger’s point, brought to bear on the question of anagenesis: it is the quantum mechanical rather than the statistical laws of nature, on which the appearance of forms is primarily dependent.
To a point, one might argue that it is the third law of thermodynamics, according to which nature approaches perfect order or zero entropy at absolute zero temperature, rather than any extrapolation from the second law, including Prigogine’s theorem, which underlies the possibility of anagenesis. For the second law and its extensions do not specify whether matter is quantum mechanical or made of little billiard balls, and it has been noted that it is difficult to build anything with spheres. This point is omitted by Weizsacker as well as by Rothstein. The omission may invalidate Rothstein’s argument in part, or render it incomplete, but it does not follow that his conclusion is wrong, and this conclusion is of great interest.
17. APPLICATIONS TO BIOLOGY.
From the physical viewpoint outlined above, the enormous numbers of different forms and activities of life are all manifestations of genetic material, which have arisen as the result of selection acting on mutations, from an initial condition which could arise with physico-chemical probability on a cooling-planet, viz., a condition characterized by macromolecules replicating themselves from constituents in the surrounding solution.
In what follows, an effort is made to discuss a few of the major physical factors involved in the transition from the initial to the modern condition, with emphasis on the statistical or informational interpretation of these factors. In other words, the aim is to consider some of the main types of anagenetic phenomena, from the standpoint of the physical interpretation of anagenesis. The discussion is divided into three parts: a. the evolution of exploitation of phenomena; b. the evolution of organ systems within particular phylogenetic lines; and c. the evolution of ecosystems. This
division is arbitrary, in that considerable overlapping is entailed. It is, however, suggestive o£ alternative ways to take aim.
a. The evolution of exploitation of phenomena.
i. Setting the stage: replication and amplification. The first necessity for the establishment of life was the development of the power of autocatalysis. It is not to be expected that exact replication was developed, all at once (cf. Boyden 1953, Dougherty 1955),’ but until a fairly accurate method was in force it is hardly possible to speak of life. Because of the danger of decimation from all sides,
replication had to be more or less wanton when circumstances were favorable. This has been true ever since, and it is reasonable to suppose that it always will be true so long as there is life.
It may be appropriate, then, to speak of the “principle of overbreeding as fundamental to biology; this principle is implicit in Malthus, and hence in Darwin. Overbreeding is not necessarily blind; in man, at least, conscious consideration can be given to its optimum. But even in a possible future condition of social control, some degree of overbreeding will remain necessary for survival, since the forces of decimation can never be fully anticipated.
Replication requires assimilation of matter and energy. This assimilation is mediated by proteins acting as catalysts, where we consider catalysis in a very general sense, as matter-energy transformation of any’ type, including the complex behavior of higher animals, in which the molecular agents of transformation emerge unchanged. (It is an extrapolation beyond the evidence to regard all behavior, along with biochemical pathways, as enzyme catalyzed, but this extrapolation is not unreasonable.)
Replication is fundamentally the achievement of DNA, as the only established biological autocatalyst. There may be some difficulties, nonetheless, with the question which came first, protein or DNA. The hypothesis that protein came first, supported by
the fact that it deals more directly with the environment, suggests the aforementioned possibility that DNA is “merely” a memory storage material, analogous to the magnetic banks of computers, rather than the fundamental material of life.
Be that as it may, the development of autocatalysis was clearly ·fundamental to biological ·evolution. It represents a phenomenon interpretable as negentropy localization much after the manner of inorganic crystal growth, in that there is carried out a progressive ordering of molecular arrangement. As Wright (1931) has cogently put it, replication achieves the multiplication of improbable configurations, instead of their dissipation.
The increasing number and variety of macroscopic structures and functions interposed during evolution between the autocatalytic molecules and external environment, also entail a progressive ordering of molecules. These structures and functions may be regarded as devices which enable the autocatalytic molecules to invade the macrocosm in quest of matter and energy for their replication, instead of remaining entirely dependent on Brownian bombardment. To an important extent, this construction and operation of macroscopic devices is achieved through amplification of molecular and sub-molecular phenomena.
The theory of amplification and its relation to biology have been discussed by Elsasser (1949) and Platt (1956); only a summary indication of apparent connections of this with other physical approaches is attempted here. Elsasser has distinguished two types of relation of micro- to macroscopic event: statistical and amplifier. In these terms, the macroscopic devices of living matter are in amplifier rather than statistical relation to microscopic phenomena, including DNA activity; and the increase in number and variety of amplifier relations is a major characteristic of anagenetic evolution. Elsasser’s distinction is reminiscent of Schrodinger’s, between thermodynamic and quantum mechanical law. From this standpoint amplifier relations appear as macroscopic manifestations of quantum mechanical rather than thermodynamic (statistical) laws, whence their evolutionary rise is interpretable as gain of molecular order, negentropy, or information.
ii. Diffusion, convection, and conduction. This series of increasingly highly ordered mechanisms of molecular motion, which grade into one another, has been entailed in the evolution of the transport of several and probably all materials required for life in one kind of organism or another–respiratory gases, cell nutrients, wastes, electro- chemical stimuli–and accordingly is implicated in the evolution of virtually all organ systems. To some extent all these processes are utilized in all living organisms. At the sub-cellular level there is perhaps little to distinguish even the bacterium from the mammal in regard to the relative utilization of the more highly ordered mechanisms. Whereas diffusion still plays the fundamental role which it may be assumed to have played since the origin of life in bringing molecules of raw materials to the site of their utilization, it is becoming increasingly clear that quite highly ordered conductive mechanisms also play important roles in cell metabolism. The structure of the mitochondrion, for example, points strongly to this interpretation (in some bacteria it may be that the cell membrane corresponds in organization and function to the mitochondrion).
At the supra-cellular level, however, there is a marked tendency for forms of increasing level of organization to utilize conduction in increasingly sophisticated ways. This is evident in ontogeny, and has doubtless occurred likewise in phylogeny. Thus we find exchange of respiratory gases, for example, carried out by means of diffusion alone in some lower invertebrates, such as acoel flatworms, while conductive mechanisms (water or air passages, together with circulatory systems) of increasing complexity are to be found in various representatives of the higher phyla such as chordates.and molluscs.
A condition in which diffusion is supplemented by convection rather than conduction is indicated, for example, in the structure of nematodes. Indeed, the negentropy levels of supra-cellular transport mechanisms point to a reinforcement of the presumption that the lower forms represent earlier conditions of life, it being arguable from the principle of continuity in the origin of forms that diffusion was employed first, convection and conduction later supplementing it, in the evolution of all types of transport in living organisms.
iii. Translatory motion. This class of phenomena includes conduction as a special case. There may be included translation of the whole body of an organism, of its parts in relation to one another, and of populations of organisms as in the migration of birds, whales, and other forms. Also not to be left out of consideration are the translatory and rotatory phenomena of man-made machines, these being ultmately gene-catalyzed like the others. All these are
low-entropy phenomena, in that they’involve an ordered motion of the molecules of the translated body with the same or functionally related velocities, in contrast to the high-entropy phenomenon of heat, which involves random motion of the molecules with respect to one another. The presumption from the statistical standpoint is that
life began without translatory motion other than Brownian, that the phenomenon appeared and was exploited for its adaptive advantage some time after replication was established. It is, in short, highly improbable that the naked genes moved under their own power at the beginning. Translation presumably became possible with the ordering of molecules into linear arrangements which were either
contractile in themselves or translatable with respect tb one another.
Initially there need have been no power of direction in order for translation to be advantageous, for it would have’ enabled individuals in a nutritionally exhausted region better to escape to richer regions of the soup, simply by increasing their range. Tropistic direction of motion would of course tend to have been advantageous whenever it appeared.
From such presumed beginnings, translatory motion has been exploited in countless ways by living matter. There exists today everything from the motion of flagellated bacteria, in some instances little more developed than what has been postulated here as the initial stage of exploitation, to the running of the cheetah, swimming of the dolphin, flight of the bee and the swift, and the remarkably highly coordinated translatory movements of the brachiating gibbon, the hummingbird, the octopus, and the seahorse, to say nothing of human athletics.
The homeostatic interactions of body parts in relative translatory motion, all coordinated in the translation of the organism as a whole and sometimes of populations, are such as to defy description in the more complex cases. All are manifestations of the low-entropy phenomenon of translation, however. Even so, it should not be forgotten that this exploitation is not a necessary condition for
evolutionary success, it being reduced or absent, except in the form of conduction, in many sessile animals and in plants.
iv. Sensory information. There has been an evolutionary trend toward more refined reception of inpui signals and toward increasingly efficient responses to them. The original naked genes could hardly have had sensory powers, unless differential response to atomic configurations of impinging molecules involved it in a rudimentary way. In contrast, human experience, for example, involves the reception of electromagnetic radiation with fine distinction of intensity, direction, and wave-length over the limited visible range, and outside that range through the mediation of electronic instruments. A similar power exists for the analysis of sound-wave information, and lesser but still appreciable chemo-sensory and mechanico-sensory powers also exist.
It is noteworthy that so much information impinges on a given point in nature. All the richness of a tropical vista, for example, is passed through the pupil ot the eye, and actually through a much smaller area, as can be shown with a pin-hole camera. Moreover, the stars of the galaxy are perceivable by the same pupil at night, and this information is also present with that of the tropical vista,
being merely temporarily swamped out by local electromagnetic
noise in daylight. Even information from distant galaxies impinges at the same place, as we know from the great telescopes, though the eye alone cannot detect it. Auditory information at a given locus near the earth’s surface is similarly rich, although its sources are much more localized.
It is as if, in the limit, the whole cosmos were represented at every point within it, by actual information carried energetically, largely in the form of electromagnetic radiation; and the evolution of life has involved a gradual improvement in ability to perceive this information and respond to it. There is no reason to suppose that this process is finished. It is defensible that increasingly accurate and wider reception of information has been advantageous in the evolution of at least the higher animals, in virtually all eras, and will continue to be so. With the increasing complexity of ecosystems there has been, moreover, an increasing amount of information in the local visual field, and presumably an increased adaptive advantage in the improvement of perception of this information.
Increasingly effective response is obviously also a necessary factor in adaptation. Presumably, improved perception and response have always been coupled to effect adaptive advantage, although perhaps never perfectly. A causal relation is indicated; the organism has latent capacities of differentiated response to information of a type received for the first time in phylogeny; otherwise there could be no advantage in its reception.
The concept of preadaptation helps to understand this situation. The development of an accurate camera-eye on land, for instance, while advantageous in food-capture, and escape therefrom, would automatically register the stars on the retina at night. Such an eye may have been developed hundreds of millions of years before any effective response to star information was made in the phylogenetic line leading to man; i.e., the mammalian or even the tetrapod eye was long preadapted to the study of stars. Celestial navigation by birds may represent an earlier utilization of star information, but the same principle applies. Moreover, the factors behind the ultimate human use of this information probably include food-gathering. Qualitative use for orientation in hunting and foraging probably preceded the quantitative use for timing the planting of crops, which paved the way for the development of astronomy.
b. The evolution of organ systems.
The viewpoint outlined in the above discussion of the evolution of exploitation of phenomena can in principle be applied to studies of the evolution of particular organ systems in a given lineage. The collective result of such studies would be a negentropic or informational description of phylogeny, in which successive “inventions” would be viewed as increasingly improbable devices for exploitation of increasingly improbable phenomena. A limitation on the feasibility of studies of this nature is put by the fact that macroscopically distinct advances in the negentropy level of devices and of corresponding exploited phenomena evidently require very long time periods, and in retreating to the times of distinctly more primitive conditions of a given system, phylogenetic relationships tend to become uncertain. This point was discussed in section 8, in terms of the long time intervals since the appearance of the deeply foundational distinguishing characters of high ranking taxa. I suggest that the “foundationality” of characters is in inverse relation to the sum of their structural negentropy and the negentropy level of the matter-energy transformations which they effect. There then arises a possibility for quantification of the concept of foundationality.
The following brief consideration of a few examples is intended to illustrate the negentropy or informational approach to phylogenetic topics.
i. Phylogeny of the circulatory system in chordates. The sequence discussed above–diffusion, convection, and conduction–recurs in the early stages of evolution of this system., with rising negentropy level of exploited phenomena. The primitive condition, in which there exist no vessels and transport is dependent on diffusion, is not known in chordate phylogeny, but is inferred to have occurred in pre-chordate ancestors on the basis of what is found in some lower invertebrates, like flatworms. The same may be said of a subsequent condition characterized by cavities in which transport 1s aided by the supplementation of diffusion with more or less turbulent motion of internal fluid.
The simplest systems found in chordates proper already exhibit conduction through vessels: 1n the cephalochordates there occurs one-way flow maintained by waves of contraction along a number of the vessels, while in the urochordates there occurs the interesting condition in which contractility is localized in a “heart” (specialized contractile vessel), but the heart periodically reverses the direction of flow. A negentropic advance over both these systems is marked by
the two-chambered heart with one-way valves, eliminating back-flow, found in cyclostomes and fishes.
A further advance is marked by the double circulation, pulmonary and systemic, found in tetrapods. The imperfect separation of the two circulations in amphibians and most reptiles is a partial step, in which a degree of convection with some mixing of oxygenated and deoxygenated blood occurs in the heart; it should be noted, however, that the flow is very smooth and the mixing remarkably little. The complete separation of the two circulations by means of the four- chambered heart, in birds and mammals, completes this step. This double circulation system can be operated at higher pressure, especially of the systemic arteries–hence with
increased rate of blood flow; it accordingly exploits more highly improbable phenomena and so marks a negentropic gain. A remarkable degree of fine-structural adaptation of the mammalian heart, and probably also of other vertebrate hearts, assures minimum convection in the passage of blood, adding to the phenomenological improbability. To a considerable extent the vessels also appear to be engineered for streamlined flow, judging from the stacking of red cells in their transport through capillaries, for instance.
Accompanying the increase of phenomenological negentropy, as it were, is an obvious increase of structural negentropy of the system, as evidenced by a glance at the circulatory system of a mammal in contrast to that of, say, Amphioxus. In addition, there are numerous feed-back mechanisms mediated by nervous and endocrine’connections, especially in the higher vertebrates. The result is a notoriously intricate example of the organ systems of
living things, reflecting a relatively high negentropy. Thus negentropic considerations seem to be in general accord with the known facts and the interpretive consensus as to the phylogeny of the circulatory system in the chordates, and they offer some illumination of this phylogeny.
ii. Phylogeny of vision. The organization of the vertebrate eye presented one of the classic difficulties which Darwin took special pains to try to account for in terms of natural selection. Although the phylogeny of the vertebrate eye is little known, some of the key events can be surmised from the comparative anatomy of forms in other phyla (cf. summary in Rensch 1959:278). The molluscs reveal perhaps the most complete series of possible steps toward image formation: a nearly flat eye spot is found in Solen, a shallow optic cup in Patella, a deep cup approaching pin-hole camera design in Haliotis and Nautilus, a spheroid camera eye with lens in Murex, and an elaborate camera eye comparable to that of vertebrates in Octopus. The discovery of series of this nature has substantiated Darwin’s general argument, by revealing stages such that it is possible to visualize the action of selection leading naturally from one to the next, toward improved image formation.
Like the stages in the evolution of the circulatory system, the presumed stages in the evolution of the eye are interpretable as steps in structural formation of, and exploitation of increasingly improbable or high negentropy states of matter-energy. Whereas structural negentropy can be viewed and probably ultimately analyzed in the same way for all organ systems as well as for’ the’ organism as a whole, the exploited phenomena are considerably different from one organ system to another, and to see them in similar negentropic terms may call for rather different approaches.
Thus, while circulation exhibits the successive exploitation of diffusion, convection, low and high pressure conduction, the evolution of vision is perhaps best viewed as a continuously improved extraction of electromagnetic information already available in every space element where an eye might be put (see previous discussion).
In the evolution of vision there is to be considered, in addition to image formation, the factors of spectrum and intensity perception. The perception of spectrum has had an evolution leading to color vision in some lines, and that of intensity has had an evolution leading to the reported perception of single quanta through the dark-adapted human eye. Less can be surmised as yet about the evolutionary steps in spectrum and intensity perception than in image formation, but an informational interpretation is perhaps rather simpler in the former cases.
Gain in information extraction capacity with respect to intensity is primarily a-matter of improvement of sensitivity of the eye, and reaches a limit with the perception of single quanta. Complications are introduced secondarily with the problem of adaptation to background intensity level, and it is more doubtful that evolution has anywhere approached a limit in this regard. With respect to spectrum, gain in information extracting capacity has involved first, extension of the width of the perceivable spectrum, and second, discrimination of wavelengths (color vision). The first is a matter of “bandwidth” of the eye as a receiver–the wider the band the more information receivable–while the second is a matter of sharpness of’
tuning. The eye with color vision, unlike the radio, tunes in on every station on the band at once, so to say, with remarkably .little confusion of wavelength information; .it does not, however, deal with informative modulations of signal frequencies to the same degree, as does the radio.
The evolution of image formation is perhaps less readily interpretable in terms of information extraction than are the other factors, but in principle the difficulties appear to be no greater. In engineering terms, the image forming ability of the eye corresponds to determination of direction by receiving antennae of all received signals at once. It may . be surmised by analogy with the development of radio and radar, as well as from eye-ser~es such as that of mollusks described above, that image formation began with something like the ability to distinguish light from darkness, and that further steps involved the distinction of light-in-front, light-above from light-below, objects-left-and-above, objects-right-and-below, etc. The number of directional distinctions which a modern camera eye can make could probably be expressed without great difficulty in terms of the amount of directional information which it is capable of extracting from its field of vision. The suggested evolutionary stages rather obviously entail an increase in this amount.
The special visual adaptations of certain forms such as the hawk, owl, or tarsier, for example, offer possibilities for the extension of this kind of analysis to higher negentropic phenomena of vision. Some surprises have come from the analysis of vision in the bee, including the remarkable discovery that bees detect perimeters better than shapes; this marks an exploitation of information in the visual field which is quite foreign to us. Watching ‘a songbird, for instance, flit errorlessly through shrubbery full of obstacles, suggests that there may be many more surprises of this kind to be discovered. Their discovery might be abetted by informationa1 considerations.
. Resolving power of the better camera eyes, being already near the 1imit for waelengths of the visible spectrum, is perhaps not likely to undergo much further evolutionary gain. Further increases of information detecting capacity are more likely to have to do with perception of relations among objects seen, past and present. There can be no question that this kind of improvement has long been an important aspect of the evolution of vision, especially in human evolution. Greater difficutties probably attend the informational analysis of this factor than the others.
iii. The phylogeny of translatory motion. It has been noted above that life has presumably evolved from a condition of naked genes entirely at the mercy of Brownian motion, to one in which’genes are surrounded by a wealth of structural and functional inventions permitting an extraordinary variety of translatory motions. With respect to structural negentropy, these inventions have to do largely with evolutionary differentiation of muscular systems, in close and intricate association especially with evolutionary differentiation of skeletal and nervous systems, but also of other organ systems. Increase in the negentropy of exploited phenomena, on the other hand, is reflected in increasing coordination of movement of body parts of individuals, and of individuals in relation to one another, interpretable as ever more improbable ordered molecular movements.
Of special interest is the phylogeny of translation in man, where perhaps .the greatest variety o( translatory phenomena within a single species is to be witnessed. This phylogeny includes, in addition to the evolution of means of translation of the individual and the evolution of translatory motion of body parts, especially the hands, also the evolution of external transport and exchange of materials, from individual to individual and population to population. Development of the latter branch of the subject would lend a negentropy interpretation to economic progress, and other changes in human history might be treated in a similar way.
Rothstein (1958) has indicated how to look at certain human activities from the standpoint of information theory. Extension of the same approach to economic and other aspects of human history, with special reference to negentropy gain during their development, is a ready generalization of Rothstein’s thought. There would be advantage in the linkage of the social sciences, as well as evolutionary biology, with a unifying physical concept.
c. The evolution of ecosystems.
If attention be focussed on the balance of nature during
evolution, rather than on particular classes of phenomena or phylogenetic problems, it is rather obvious that the increasing number and variety of structures and functions of living matter, with their increasing improbabilities of state, entail rising negentropy of the systems in balance, or ecosystems, including the biota as a whole. Again, no contradiction of the second law of thermodynamis is necessary, since the ecosystem is an open system. An application of information theory to ecosystems, with calculations, has been made by Patten (1959), although application to the anagenetic evolution of such systems is not much developed by him, the emphasis being on ecological succession by forms already in existence. His work suggests that it may be not entirely fanciful to pursue quantification even of ecology and its evolution through information theory. It is to be expected, however, that a high degree of precision is unattainable in principle, because of the point mentioned before, that systems highest in the hierarchy of levels of organization equal or transcend the negentropy level of the inquiring mind.
It may be justifiable to emphasize here some qualitative
aspects of the negentropy increase of ecosystems. I am indebted to Dr. D. P. Abbott for .suggesting some of the points considered.
To begin with, there has been an increase with time in the number of individuals and of species participating in the biota. The ptinciple behind the negentropy increase which accompanies simple replication of macromolecules–in Wright’s terms, as noted earlier, replication achieves the multiplication instead of the dissipation of improbable states–is also behind the rise in negentropy associated with ihe increase in number of species. It has been estimated that the number of species existing at anyone time on the planet has doubled approximately every 80 million years (Cai1leux 1954). The increase in species number has undoubtedly been an important factor throughout most of the history of life. However, there would appear to be a mathematical limit to the possible number of species which could coexist on a finite planet. Moreover, the recent effect of man has been to decrease the number of existing species of some groups, if not all. It is therefore only with reservations that one can look to this factor as being involved in an important way in future increases in the negentropy of the planetary ecosystem.
Associated with the increase in number of species there has been an increase in the total area and in the types of environment occupied by living matter. Although invasions may have begun with individuals of even a single species, they have led to occupation by communities and by inc’reasingly complex balanced ecosystems. The occupation of the land is a familiar case in point. Once again, of course, there are probably limits to negentropy gains from such sequences of events, posed by the finiteness of the planet. The emerging possibility of colonization of other planets, however, could keep open the door to unlimited extension of the operation of this factor, and perhaps also of the first.
The invasion of new areas, such as the land, has typically required the evolution of more complex forms, because of the relative difficulty of maintaining life processes in the rtew environment. The elaboration of structures and correlated functions in the invaders (e.g., apparatus for air breathing, limbs adapted to terrestrial locomotion, etc.) entail negentropy gains at the organismic as well as ecosystem level in the invasion process.
In relation to this point, it may be surmised that, with the general increase in buffering between genes and fluctuating environment which evolution has produced, there has been a tendency toward increased stability, with decreased risk of extinction, of ecosystems as well as of organisms. Thus a short drought or a bad storm could be expected to have wreaked much greater havoc on a land community in the Devonian than it would today. Protective structures and feedback regulatory mechanisms reflecting negentropy gains are again behind this effect.
The formation of communities in new domains, and the subsequent increases of ecosystem complexity there, entail the replacement of purely geochemical processes by bio- geochemical cycles. Therewith are entailed also increases in the volume of the biosphere, in the number of steps and feedback mechanisms in matter and energy transformation chains or webs, and in the numbers and quantities of different materials going through biogeochemical cycles (complexity of the “ecomix”). Patten’s (1959) calculations of increase of negentropy capture in ecological succession,toward a maximum capture by climax systems, suggests the generalization that the same may be true on the macro-evolutionary scale at issue here, although there are no terminal climaxes on this scale.
As noted earlier, the structures and devices built by organisms, including spiderwebs and skyscrapers, may be looked upon as being ultimately gene-catalyzed in essentially the same sense as are the biochemical pathways in the mitochondrion. The negentropy of these structures and devices should be included in that of the biota as a whole under this view. Such transformations of the environment then represent another factor reflecting negentropy increase in the evolution of organisms and ecosystems. It is for this reason that, if highly organized structures like our cities were to become visible in an approach to the surface of another planet, one would strongly suspect the presence of life there, albeit not necessarily life based even on the same chemistry as our own. Indeed, it is this inference which was made long ago in postulating life on Mars, because of the supposed appearance of “canais” on its surface.
Factors other than negentropy accumulation of course enter into the evolution of ecosystems. For example, Sahlins (in Sahlins and Service 1969) has recently stressed increase in total harnessed energy as a criterion of evolutionary progress, with special reference to measurement of the advance of human cultures. In a similar vein, there is something to be said for, say, horsep.ower production per capita as an index of cultural level, or at least of technological level. More generally, power output (energy transformation per unit time) per unit biomass, including power output directly from metabolic processes, would provide a standard applicable to any species, or even any ecosystem. An increase in this power output might be taken as a criterion of evolutionary advance, of species or ecosystem. Still more fundamental, perhaps, would be a measure of power output per unit genetic material, again applicable in principle to culture, species, or ecosystem. The negentropic viewpoint, however, focusses on an important element overlooked in the consideration of power output alone. The number and variety of structures and matter-energy exploitations, and their degree of improbability, are aspects of
the evolutionary level of a species or ecosystem as important as power output, if not more so. The measure of rate of negentropy capture being even more difficult than that of power output, if possible at all, it is unlikely that this rate can become a practical index of evolutionary advance, at least in the foreseeable future. The principle, however, has some significance even as a rough idea, perhaps not least because human values appear to be more closely linked with negentropy capture and exploitation than with power
output (cf. Rothstein on skill, art, science, and values, for instance).
Presumably, then, the structural negentropy of organismic devices, together with the negentropy of energy exploitations effected by them, are reflections of the information content of the genetic material. From this standpoint, genetic information is what we value, or is at least an aspect of what we value. This must include less than the total information in the human gene pool, insofar as errors and misinformation therein are sometimes worse than valueless; but it must also include more, insofar as human life depends upon the information content of other gene pools of the biota, and on the information or negentropy of the matter-energy on which life feeds. In general, it may be preferable to think of values as biotic rather than human. The value of a biota might then be conceived as partially measured by the information content of its genetic material, a principle of possible utility in “comparative exobiology” (cf. Lederberg 1960).
LITERA TURE
Bader, R. S. 1958. Similarity and recency of common ancestry. Syst.
~. 1:184-187. Beadle, G. W.
1957. The physical and chemical basis of inheritance. Condon Lectures, Oregon State System of Higher Education, Eugene.
Beerbower, J. R.
1953. The chronofauna and quantum evolution. Evolution 1: 178.
Bigeiow, R~ S. 1956. MonQphy1etic classification and evolution. Syst.
Zool. 5:145-146. Blum, H. F.
1951. Time’s arrow and evolution. Princeton University Press, Pr1nceton. (Ed. 2, 1954).
Bock, W. J.
1959.
Bohr, N. 1933.
Preadaptation and multiple evolutionary pathways. Evolution 13:194-211.
Light and life. Nature 131:421-423, 457-459. A l s o i n : B o h r , N. Atomrc-~sics a n d human knowledge. Wiley, New York, 1958, W. 3-12.
Boyden, A. A.
1953. Comparative evolution with special reference to primitive mechanisms. Evolution 1:21-30.
Branson, H. R.
161
1953. A definition of information from the thermodynamics of irreversible processes. In: Quastler 1953:25- 40.
Bray, J. R.
1958. Notes toward an ecologic theory. Ecology 39:770- 776.
Brillouin, L.
1949. Life, thermodynamics, and cybernetics. Amer. Scientist 37:554-568.
1950. Thermodynamics and information theory • .Amer. Scientist 38:594-599.
1951. Information and entropy. J. Applied Phys. 22:334- 343.
1956. Science and information theory. Academic Press, New York-.-
Brough, J.
1958. Time and evolution. In: Westo1l, T. S. (Ed.), Studies on fossil vertebrates. Athlone, London, p p . 16-38:”
Brown, W. L., Jr.
1958. General adaptation and evolution. Syst. Zool. 7:157-168.
Cai11eux, A. 1954. How many species? Evolution ~:83-84.
Darwin, C.
1859. On the origin of sgecies. Reprint of first edition, W itts, London,-r95 •
[Note: Quotations in the text are from this edition, which has the same pagination as the 1951 edition of the Philosophical Library, New York. The essential content of the quoted material is the same in Darwints sixth and last edition of 1872J
De Beerf G. R.
. 1949. 1958. The physical foundation of biology. Pergamon,
162
1930. Embryo1og¥ and evolution. Oxford University Press, Ox ord.
1958. Embryos and ancestors. Ed. 3. Oxford University Press, Oxford.
DeGroot, S. R.
1952. Thermodynamics £f irreversible processes~ North- Holland, Amsterdam, and Interscience, New York.
Dobzhansky, Th. 1937. Genetics and the or1g1n of species. Columbia
Unl.versl.tnress'. New York. J.941. .!.£!s!. Ed. 2.
1951..!.£!s!. Ed. 3: 1959. Evolution of genes and genes in evolution. In:
Cold Spring Harbor Sympos. Quant.~. 24:15-30. Dodson, E. O.
1960. Evolution: process and product; Reinhold, New Y ork.
Dougherty, E. C.
1955: Comparative evolution and the origin of sexuality. Syst. Zool. i:145-l69,190.
Eiseley, L. C. 1958. Darwin's century. Doubleday, New York.
Elsasser, W. M.
Quantum mechanics, amplifying processes, and living matter. Proc. Utah Acad. Sci., Arts, and
Letters (1948-49),26:89=Tl~ - - - .
.
New York.
1961. Quanta and the concept of organismic law. d. Theoretical ]i£!. 1:27-58•
– ,
Emerson, A. E.
163
1960. The evolution of adaptation in population systems. In: Tax 1960:307-348.
1961. Vestigial characters of termites and processes of regressive evolution. Evolution 15:115-131.
Flower, R. H.
1955. Saltations in nautiloid coiling. Evolution 9:244-259.
Goldschmidt, R. B.
1940. The material basis of evolution. Yale University Press, New Haven. Repr~nted 1961, by Cooper Square, New York.
1952. Evolution, as viewed by one geneticist. Amer. Scientist 40:84-98.
1955. Theoretical genetics. University of California Press, Berkeley.
1960. In and out of the ivory tower. University of Washington Press, Seattle.
Hanson, E. D. 1961. Animal diversity. Prentice-Hall, Englewood
c1iffs,~ew J e r s e y . 1954. The old~st dated minerals of the.Rhodesian shield.
Nature 173:612-616. Holmes, S. J.
1944. Recapitulation and its supposed causes. Quart. Rev. Bio1. 19:319-331.
i948. The principle of stability as a cause of evolution. A review of some theories. QUart. Rev. !!i£!.. 23: 324-332.
Holmes, A.
.. 1942. Evolution: the modern synthesis. Harper, New
Y ork.
164
1957. The three types of evolutionary process. Nature 180:454-455.
Hyman, L . H. 1959. The invertebrates. V. Smaller coelomate groups.
~raw-H1ii, New Y o r k . Kavanau, J. L.
1960. On the or1g1n of life: letter to the editor.
Science 131:1682.
Kerkut, G. A. 1960. Implications of evolution. Pergamon, New York.
Lederberg, J. 1960. Exobiology: Approaches to life beyond the earth•
. Science 132:393-400. Lewis, E. B.
1951. Pseudoa11e1ism and gene evolution. Cold Spring Harbor Sympos. Quant. Bio!. 16: 159-1~
Lewis, H., and P. H. Raven
Huxley, J. S.
1958. Mayr, E.
1,942.
1960.
Rapid evolution in Clarkia. , Evolution 12:319-336. -r-
Systematics and the or1g1n of species. Columbia Un1vers1ty Press~ew Y ork.–
The’ emergenc~ of evolutionary novelties. In: Tax 1960:349-380.
Meyer, A. W.
1935. Some historical aspects of the recapitulation idea. Quart•.~. ~. 12:379-396.
165
Michener, C. D., and R. R. Soka1
1957. A quantitative approach to a problem in classifI- cation. Evolution 11:130-162.
Patten, B. C.
1959. An introduction to the cybernetics of the ecosystem: the trophic-dynamic aspect. Ecology 40:221-231
Petrunkevitch, A. 1952. Macroevolution and the fossil record of
Arachnida. Amer. Scientist iQ:99-l22. Platt, J. R.
1956. Amplification aspects of biological response and mental activity. Amer. Scientist 44:180-197.
Quastler, H. (Ed.)
1953. Essays on the use of information theory in biology. Un1vers1ty-of lTIinois Press, Urbana.
Reed, C. A. 1960. Polyphyletic or monophyletic ancestry of mammals,
or: What is a Class? Evolution 14:314-322. 1947. Neuere Probleme der Abstammun~slehre. Die
transspez1f1sche~01ut10n. • E n k e , S t u t t g a r t . 1954. ~. Ed. 2.
1959. Evolution above the species level. Columbia University Press~ew York.
Rothstein, J.
1958. Communication, organization, and science.• Falcon’s Wing Press, Indian H1lls, Colorado.
Rensch, B.
166 Sah1ins, M. D., and E. R. Service
1960.
Evolution and culture. University of Michigan Press, Ann-xrbor.
Schindewo1f, O. H.
1950. Grundfragen der Pa1eonto1ogie. E. Schweitzer- barthsche, StUttgart.
Schma1hausen, I. I. 1960. Evolution and cybernetics. Evolution 14:509-524.
Schrodinger, E.
1944. What is life? Cambridge University Press, CambrIdge:-
1958. Mind and matter. Cambridge University Press, Cambridge.
Shull, A. F. 1936. Evolution. McGraw-Hill, New York. 1951. ~. Ed. 2.
Shumway, W.
1932. The recapitulation. theory. Quart.~.~. 7:93-99.
Simpson, G. G. 1944. Tempo and mode in evolution. Columbia
On1verSIty-pres~ New Y ork. 1949. The meaning of evolution. Yale University
~ss, New Haven. .1953. The major features of evolution. Columbia
Univers1ty Press, New York.
1959a. The nature and origin of supraspecific taxa. In: ColdSpringHarborSympos.Quant.~.24:255-271.
Simpson, G. G.
167
1959b. Mesozoic mammals and the polyphyletic origin of mammals. Evolution 13:405-414.
1960a. The history of life. In: Tax 1960:117-180.
1960b. Diagnosis of the classes Reptilia and Mammalia. Evolution 11:388-392.
1961. Principles of animal taxonomy. Columbia Univers1ty Press, New York.
Simpson, G. G., C. S. Pittendrigh, andL. H. Tiffany 1957. Life. Harcourt, Brace, New York.
Singer, C.
1950. ~ history of biology. Revised ed.Schuman, New York.
Sinsheimer, R. L.
1957. First steps toward a genetic chemistry. Science 125:1123-1128.
Stebbins, G. L., Jr.
1950. Variation and evolution in plants. Columbia University Press, New York.
Stephens, S. G.
1951. Possible significance of duplication in evolution. Advances in genetics i:247-263.
Straus, W. L., Jr. 1958. Evolutionary terminology. Science ~:22.
Tax, S. (Ed.)
1960. Evolution after Darwin. I. The evolution of life. University of Chicago Press, Chicago.
168
van Va1en, L. 1960. Therapsids as mammals. Evolution !i:304-313.
W addington, C. H. 1960a. Evolutionary adaptation. In: Tax 1960:381-402. 1960b. The ethical animal. Allen and Unwin, London.
Weir,J. A.
1946. SparinR genes for further evolution. Iowa~. Sci. 53:313-319.
von W eizsacker, C. F. 1949. The history of nature. Tr. by F. D. Wieck.
UniverS1 ty orChicago Press, Chicago’. Wilson, H. V.
1941. The recapitulation theory or biogenetic law in embryology.~.Nat.75:20-30.
Wright, S.
1931. Statisticaltheoryofevolution.~.Stat.~., Mar. suppl.:201-208.
1959. Genetics and the hierarchy of biological sciences. Science 112:959-965.
Yockey, H. P. (Ed.)
1958. Symposium ~ information theory in biology. Pergamon, New Y ork.